Federal Vaccine Advisory Committee Update—ACCV and NVAC
The National Vaccine Information Center (NVIC) regularly monitors meetings held by federal vaccine advisory committees, including the Advisory Commission on Childhood Vaccines (ACCV) and the National Vaccine Advisory Committee (NVAC). Both ACCV and NVAC were created under the National Childhood Vaccine Injury Act of 1986 and committee members are appointed by the U.S. Department of Health and Human Services (DHHS) to usually serve two to four-year terms.
The ACCV is charged with providing oversight and giving advice to DHHS on the federal Vaccine Injury Compensation Program (VICP) that Congress designed in the 1986 law to serve as an alternative to vaccine injury lawsuits against vaccine manufacturers and doctors.
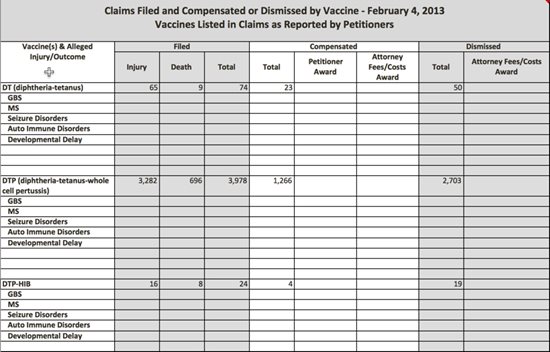
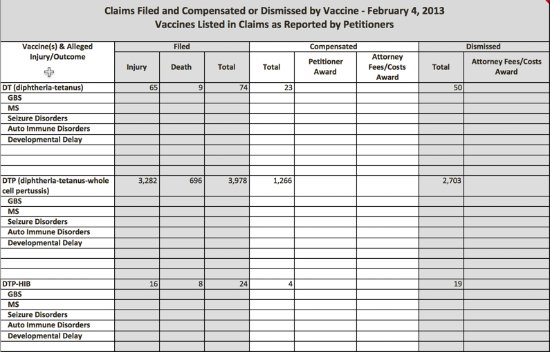
During ACCV meetings, the U.S. Department of Justice (DOJ) and Division of Vaccine Injury Compensation (DVIC) present quarterly data on vaccine injury claim petitions filed and the length of time each petition took to process; petitions that result in federal compensation; how many cases were adjudicated by concession, settlement or through a U.S. Court of Claims decision; and how many cases were dismissed.
The Public Right to Vaccine Injury Information—The Nut Shell
In recent years, information about compensated vaccine injury claims published in the VICP Data and Statistics Report has changed—but not for the better. This report prepared by DHHS officials continues to provide limited insight into vaccine injury and death awards, despite the fact that federal law states the public has a right to additional information,1 much of which is presented in bits and pieces during ACCV meetings.
Under the 1986 law, the public is entitled to the following information about VICP vaccine injury and death petitions:
- the number of petitions filed with the VICP and their disposition, the dates of when vaccine-related injuries and deaths occurred;
- the types and amounts of awards; and,
- the length of time for the disposition of petitions;
While DHHS gives some of this information to the ACCV on a quarterly basis, the information is not as meaningful as it could be because of the way it is presented in ACCV meetings and on the VICP’s website. There is no ability for the public, or the ACCV for that matter, to connect the dots between vaccine injuries compensated by vaccine and condition over time, or to determine the reasons and any associated trends on why petitions are dismissed and claimants are denied compensation.
Transparency with the Public Not a Priority
NVIC has requested greater transparency in reporting of compensated vaccine injury claims data since 2013.2 In 2014 NVIC participated in a meeting at the ACCV’s invitation, to increase public access to vaccine injury data in accordance with the 1986 law.3
Below is an example using fictitious data of what is possible with existing vaccine injury claim data, the majority of which is presented during ACCV meetings, and submitted to the ACCV’s Process Workgroup for consideration.
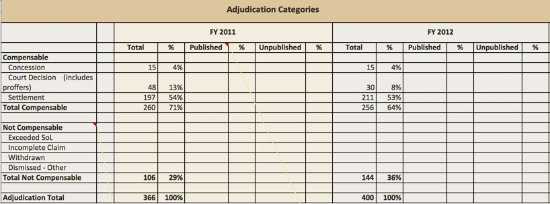
On behalf of NVIC, a request was made by ACCV to the Division of Vaccine Injury Compensation (DVIC) at DHHS to investigate the feasibility for DHHS to fully integrate detailed vaccine injury claim information presented to the ACCV into the Data and Statistics Report published on VICP’s website. The response by DVIC Director Dr. Melissa Houston to the ACCV’s request was that the information currently provided by DHHS, DOJ and the U.S. Court of Claims met legal requirements and was adequate.4 In essence, the response by DHHS leaves the public with two options:
1. visit and search multiple websites to compile the data on your own, if you know where to find it; or
2. submit a Freedom of Information Act (FOIA) request to DHHS, which often takes months and results in the demand that a fee must be paid to get the information.
Why is more detailed data about compensated vaccine injury claims important? Because the ACCV uses this information to guide its recommendations to DHHS about vaccine safety research priorities and which vaccine injuries should be added to the Vaccine Injury Table that used by DHHS, DOJ and the U.S. Court of Claims to evaluate vaccine injury claims. It could also inform ACCV recommendations for changes to the VICP process to facilitate the awarding of federal compensation to those who are injured or die from vaccine reactions.
In short, more detailed information about compensated vaccine injury claims would better inform ACCV recommendations, as well as provide transparency by living up to the spirit and intent of preventing vaccine reactions, injuries and deaths that was intended by Congress when creating the 1986 law.
Incomplete Vaccine Injury Claim Data the Norm
During the ACCV’s last meeting in June 2015,5 NVIC once again publicly raised concerns about incomplete reporting of vaccine injury claim data and the arbitrary manner in which DHHS publishes information in the VICPs’ Data and Statistics Report. Specifically, NVIC pointed out that data published in that report were either not current or incomplete.6
In 2013 a new table was added to the report that detailed, by vaccine, whether or not a vaccine injury claim was conceded, settled, received a court decision or was dismissed.7 While the addition provided some new insight into a claim’s ultimate disposition, the data was, and continues to be, incomplete.
Instead of the table providing data since the VICP’s inception (1988), the timeframe it now spans is from 2006 to 2014.8 Oddly, when the table was first published in 2013 it contained data from 2006 to present and was updated on a monthly basis with current data.9 It was early in 2015 that DHHS made an arbitrary decision to withhold current information on vaccine claim dispositions, though it continues to be presented during ACCV quarterly meetings.10
This truth and transparency violation was also recently noted by award winning investigative journalist Sharyl Attkisson11 The response that Ms. Attkisson received from the DHHS Health Resources and Services Administration (HRSA) during her inquiry about posting of incomplete VICP data was that “an internal decision was made to ensure that all internal data was consistent… and to update the chart only when all relevant data was available,”.
The truth and transparency concerns posed by NVIC and Ms. Attkission raise the following questions:
- Why is no data prior to 2006 on vaccine injury claim disposition (categorized by vaccine) made available to the public by DHHS?
- Why has DHHS discontinued monthly updates of vaccine injury claim disposition (by vaccine), as was done previously? Why is current data no longer considered relevant?
- Why does DHHS refuse to integrate data presented to the ACCV meaningfully into the Data and Statistics Report?
Irrelevant Additions of Data Minimize Vaccine Injuries and Death
DHHS has added new information to the VICP’s Data & Statistics Report posted online, but that information appears to be largely irrelevant or designed to minimize the significance of vaccine injury and death claims.
For example, the public now has access to the total number vaccine doses distributed (sold) in the U.S. compared against the number of vaccine injury petitions adjudicated (both those that are awarded federal compensation and those that are dismissed or denied compensation).
There is also a new narrative that precedes the actual Data & Statistics data tables stating that the majority of vaccine injuries are mild and that serious injuries are rare. However, there is no mention of the fact that the majority of vaccine reactions, injuries and deaths are never reported to the federal Vaccine Adverse Event Reporting System (VAERS),12 or that most Americans do not even know that there IS a federally operated vaccine reaction reporting system or a federal program for vaccine injury compensation with hard deadlines for filing claims.
The new DHHS narrative also lacks transparency in that there is no mention of the significant vaccine safety research gaps reported by the Institute of Medicine (IOM) in more than 20 years of published reports, most recently in IOM reports published in 1994,13 201214 and 2013.15 The ACCV uses IOM reports to make research and vaccine injury table recommendations, thus these reports are well known by DHHS.
These new additions to the VICP’s Data and Statistics Report are not required by law, and appear to serve only one purpose: to diminish vaccine injuries and deaths and promote the idea that the vaccine injured are acceptable collateral damage for the “greater good.” Perhaps this is not surprising, given the adoption by the U.S. government of cradle to the grave one-size-fits-all mandatory vaccination policies resulting from the financial public-private partnership held between the government and the pharmaceutical industry.16 17
Clearly, DHHS is reluctant to be transparent about vaccine injuries and deaths using the excuse that providing truthful, comprehensive information about compensated vaccine injury claims to the public is “not relevant” or that they have already met their legal requirements. Instead, the business of the day is to offer a minimum of information manipulated against a backdrop of federal agencies deciding what is relevant and/or adequate, while securing maximum use of all federally recommended vaccines by every American with no hesitation or questions asked.
Feds Continue Plans to Track Down Non-Compliers
The National Vaccine Advisory Committee (NVAC) has operated since 1988 and reviews vaccine science, research, supply and policy issues and makes recommendations to the Assistant Secretary of DHHS. The NVAC recently published a new report with their recommendations on how to address vaccine hesitancy.
The report, Assessing the State of Vaccine Confidence in the United States,18 details strategies for compliance with the CDC’s vaccination schedules, but does little to address confidence as it relates to vaccine adverse event data and safety concerns held by the public.
While the report purports to measure confidence in federally recommended vaccines, the real goal appears to be to get every man, woman and child living in the U.S. to comply with Healthy People 2020 vaccination goals.19 The report documents that 20 percent of parents have low confidence in the judgment of vaccine use recommendations made by the medical trade association, American Academy of Pediatrics (AAP), as a safe and effective way to prevent serious disease and 27 percent of adults didn’t trust the vaccine use recommendations made by public health officials.
The report’s solution for this crisis of faith is the identification of those who delay or decline one or more vaccines via state Immunization Information Systems (IIS) and Electronic Health Records (EHR) that health providers have been incentivized20 21 22 to use. These records are being integrated into databases at the expense of traditional medical privacy between you and your health care provider.23
Citizens can also expect their hard-earned taxpayer dollars to be spent on yet more communications to “enhance” vaccine confidence and develop “systems to support parent and community efforts to promote” vaccines. In plain language, your friends and neighbors will be tapped to talk to you if your child is not in compliance with CDC’s vaccine recommendations that children receive 69 doses of 16 vaccines starting at birth to age 18. The report’s recommendations also extend to suggesting ways doctors can persuade adults to comply with the CDC’s adult vaccination schedule24 and get annual flu shots for the rest of their lives, along with other vaccines like shingles, HPV, Hib, pneumococcal, meningococcal vaccines.25
This government sponsored coercion is designed to continually pit citizens against each other and interfere with what is—and should remain—a personal medical decision.
Strong Arm Tactics Used to Reach Goals
Although the report noted that concerns held by parents have increased over time, the overarching premise of the NVAC’s “confidence” report is that compliance with federally recommended vaccines is the societal norm in America and anyone who opposes it should be strong armed or sanctioned for non-compliance. This premise denigrates legitimate vaccine safety concerns of Americans, who are being aggressively lobbied to get every federally licensed and recommended vaccine. Many of these legitimate concerns about vaccine safety science gaps are echoed in IOM reports.26
In addition to tracking down non-compliers, the NVAC’s road plan to meet Health People 2020 vaccination goals includes recommendations that states mandate vaccine education and informed refusal statements to make it harder for parents to exercise of non-medical (religious or conscientious belief) vaccine exemptions for their children and that school vaccine exemption data be publicized. Health insurance plans will also be encouraged to implement “on-time-vaccination” as a quality control measure so the government can use it as an indicator of vaccine confidence.
NVIC’s public comments on the NVAC’s vaccine confidence report27 have emphasized that Healthy People 2020 Goals, are aspirational goals and that strong arm tactics will not inspire public trust and demonstrate a lack of respect for medical privacy. Further, a one-size-fits-all “no-exceptions” government vaccination policy does not respect the informed consent or precautionary principles and fails to acknowledge that vaccines are no different from other pharmaceutical products—they carry a risk for injury and death that can be greater for some people and those individual risks often cannot be predicted prior to use of the product.28
What You Can Do
Ask your doctor about how your personal medical information is being used and if it is being shared with others. Before receiving medical care in a doctor’s office, you are usually asked to sign a HIPAA (Health Insurance Portability and Accountability Act) form. Take steps to protect your medical privacy, if you feel that it is being compromised.
Visit NVIC’s state pages to link to your state’s Immunization Information System (IIS) and learn more about it. Laws vary from state to state. Find out if your state’s IIS is an “opt-in” or “opt-out” system. If you do not want your vaccination status or that of your child incorporated into your state’s vaccine tracking registry and/or shared with federal health officials, find out how to prevent that information from being included in your state’s IIS.
Register for NVIC’s Advocacy Portal (NVICAP) and act to protect your legal right to make voluntary vaccination decisions for yourself and your children. NVICAP issues email alerts when vaccine exemptions are being threatened (or expanded) in proposed state and federal legislation. Portal users are put in direct electronic contact with their elected representatives and receive talking points and information resources that make it easy to communicate with legislators.
Use the illustrated NVIC Reforming Vaccine Law & Policy Guide to start the conversation with your legislators and community leaders to build relationships based on accurate facts and informed consent principles. This fully referenced guide is an invaluable vaccine education tool and now is a good time to contact your legislators because they are not in session and have more time to meet with their constituents.
References:
1 The National Childhood Vaccine Injury Compensation Act of 1986, 42 U.S.C. §§ 300aa-25 -Recording and Reporting of Information.
2 Wrangham T. NVIC ACCV Public Comment, March 2013.
3 Ibid footnote 1.
4 Advisory Commission on Childhood Vaccines (ACCV) – Certified Minutes for Sept. 4, 2014, p. 2.
5 ACCV Agenda. June 4, 2015.
6 Wrangham T. NVIC ACCV Public Comment. June 4, 2015
7 National Vaccine Injury Compensation Program (VICP) Adjudication Categories by Vaccine for Claims Filed Calendar Year 2006 to Present. Aug. 13, 2013. Accessed Aug. 25, 2015.
8 National Vaccine Injury Compensation Program (VICP) Adjudication Categories by Vaccine for Claims Filed Calendar Year 2006 to 2014. Aug. 1, 2015. Accessed Aug. 25, 2015.
9 National Vaccine Injury Compensation Program (VICP) Adjudication Categories by Vaccine for Claims Filed Calendar Year 2006 to Present. Aug. 13, 2013. Accessed Aug. 25, 2015.
10 ACCV June 2015 Meeting Book, Meeting Presentations and Updates, Tab 5.1 and 5.2.
11 Attkisson, S. Govt Wipes Recent Vaccine Injury Data From Website. June 30, 2015.
12 U.S. Vaccine Adverse Event Reporting System Limitations.
13 Institute of Medicine Report – DPT Vaccine and Chronic Nervous System Dysfunction: A New Analysis. Washington, DC. The National Academies Press. 1994.
14 Institute of Medicine Committee to Review Adverse Effects of Vaccines. Washington, DC: The National Academies Press 2012.
15 Institute of Medicine Report – Childhood Immunization Schedule and Safety: Stakeholder Concerns, Scientific Evidence, and Future Studies. Washington, DC. The National Academies Press. 2013.
16 Kirby TL. Licensing and Patenting at the NIH. Office of Technology Transfer, NIH: 2013.
17 Fisher B.L. The Vaccine Culture War in America: Are You Ready?. NVIC eNewsletter. Mar. 8, 2015.
18 National Vaccine Advisory Committee Draft Report – Assessing Vaccine Confidence in the United States. March 2015.
19 Healthy People.gov. Immunization and Infections Diseases.
20 CDC. Meaningful Use and Immunization Information Systems.
21 Centers for Medicare and Medicaid Services – CMS.gov. EHR Incentive Programs. The Official Web Site for the Medicare and Medicaid Electronic Health Records (EHR) Incentive Programs.
22 Ibid footnote 18.
23 TEDx Traverse City 2014. Designing Technology to Restore Privacy. Deborah C. Peel, MD.
24 NVIC 49 doses brochure.
25 CDC. Adult Immunization Schedule. 2015.
26 Ibid footnotes 13, 14, and 15.
27 Wrangham T. NVIC Public Comment to NVAC. June 9, 2015.
28 Institute of Medicine Committee to Review Adverse Effects of Vaccines. Adverse Effects of Vaccinations: Evidence and Causality. Evaluating Biological Mechanisms of Adverse Events: Increased Susceptibility. p. 84. Washington, DC: The National Academies Press 2012.