New weight loss drug Ozempic has taken the world’s obesity epidemic by storm, but like most weight loss drugs that precede it over the past century, shedding extra pounds may not be the only outcome for consumers: Last month, the European Medicines Agency (EMA), a European Union (E.U.) agency that oversees and evaluates pharmaceutical medications, issued a safety signal, citing concerns about a possible link to thyroid cancer in people using glucagon-like peptide-1 (GLP-1) receptor agonists, including semaglutide, which are sold under brand names like Ozempic, Wegovy and Mounjaro.1
In April, the agency required drug makers who manufacture semaglutide, which include Novo Nordisk, Eli Lilly, AstraZeneca, and Sanofi, to provide supplemental documentation on the drug by July 23, 2023. Prompting the regulation was a February 2023 study that found that people using GLP-1 receptor agonists for one to three years may have an increased risk of all types of thyroid cancers.2
The demand for the drug that was approved by the U.S. Food and Drug Administration (FDA) in 2017 for the treatment of diabetes continues to outrun supply after being prescribed off-label on a mass scale for the treatment of obesity, leaving many diabetes patients frustrated as they are unable to access medication to manage their chronic condition.3
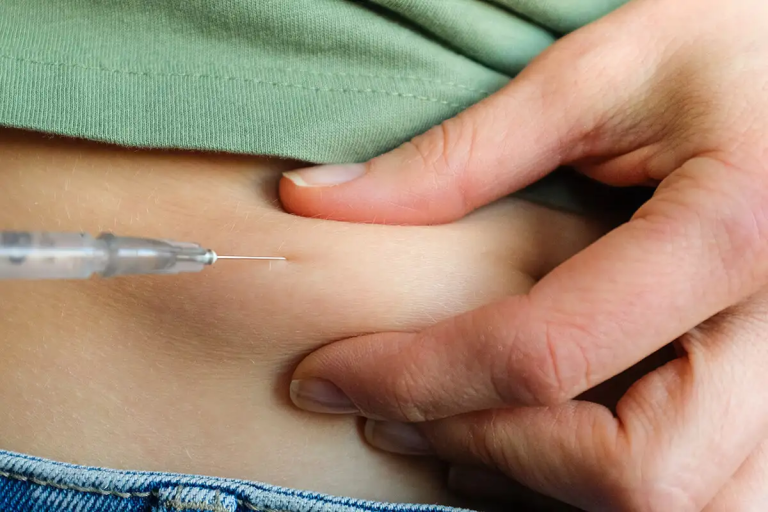
Semaglutide is a GLP medication that tricks the brain into feeling full and suppresses appetite but lost in the fine print of the weight loss miracle drug are some major side effects not commonly provided to patients being prescribed the drug, reigniting concerns about the importance of providing consumers with informed consent. Wegovy, Ozempic’s sister drug that uses the same formula at a lower dose and is also manufactured by Nova Nordisk, is the only GLP-1 drug that is FDA-approved for chronic weight management. The drugs, in addition to promoting fast and significant weight loss, can also lower blood sugar, making it a viable treatment for diabetes management.
FDA Warns Ozempic Users of Other Safety Concerns
Earlier this month, the FDA recently received reports of problems after people used off-brand, compounded versions of semaglutide, usually sold online. While they did not go into detail about the problems, they did warn that users should only use semaglutide under the direction of a licensed healthcare provider and a state-licensed pharmacy. The agency warns that compounded versions of semaglutide may contain salt forms of semaglutide—semaglutide sodium and semaglutide acetate—which have not been shown to be safe or effective. “Compounded drugs are not FDA-approved, and the agency does not verify the safety or effectiveness of compounded drugs,” the notice said.4
Doctors Warn Ozempic’s Long-Term Effects Unknown
While it’s clear GLP medications like Ozempic, Wegovy and Mounjaro are effective for weight loss, doctors warn side effects could lead to hospitalization, and that the long-term impacts of the drugs are still unknown.5
The most common side effects are nausea, diarrhea, stomach pain, constipation and vomiting, with some patients experiencing such a suppressed appetite that they become malnourished. More serious side effects include thyroid tumors, pancreatitis, changes in vision, hypoglycemia, gallbladder issues, kidney failure, and cancer.
A black box warning on the manufacturer’s insert—the highest safety-related warning intended to alert the consumer to major risks of the drug—states that semaglutide caused thyroid C-cell tumors in rodents, and that it is unknown whether Ozempic causes these tumors in humans. The latest findings from the EMA last month, however, suggest this risk is also present for humans. ¹
Liver Damage in Study Participants Halts Development of Pfizer’s New “Ozempic-Like” Pill
Pfizer has been working to develop a weight loss pill that rivals Ozempic, but when participants in one study experienced elevated liver enzymes that could indicate liver damage, the project was brought to a screeching halt. Pfizer said in a statement they will continue working on their other weight loss pill contender, danuglipron, which they assure promises noticeable results without the potential liver damage.6
Are weight loss drugs the pharmaceutical industry’s new cash cow? Pfizer CEO Albert Bourla says their new obesity pill could yield a staggering $10 billion a year. Meanwhile, Danish drugmaker and manufacturer of Ozempic and Wegovy, Novo Nordisk, currently holds the top spot for the biggest year-over-year revenue growth in 2023, according to an Evaluate Vantage annual world preview report.7 8
WeightWatchers and Noom Expand Business Models to Provide Access to Risky Weight Loss Drugs
It’s not only pharmaceutical companies that are pining for a piece of the semaglutide pie: Popular weight loss programs like WeightWatchers and Noom are also expanding their business models to offer the obesity drug to their users. In March 2023, WeightWatchers acquired telehealth platform Sequence in a $132 million purchase as a segue into making the obesity drugs available to their subscribers.9
WeightWatchers’ chief scientific officer Gary Foster said of the acquisition that the company intends to “provide the support needed to help people build livable healthy habits for the long term and manage the dietary-induced side effects often felt while taking chronic weight management medications.”9
The Dark and Checkered Past of Popular Weight Loss Drugs
From widely-used amphetamine-laden diet pills of the 1960s to the “miracle” weight loss drug fen-phen in the 1980s, the history of weight loss drugs is not only long—it’s deadly. Benzedrine and Dexedrine were choice diet pills for many in the 60s, including music icon Elvis Presley, whose longtime amphetamine use, according to one case study, contributed to his untimely death at age 42 in 1977.10
Fen-phen had all the clout in the 80s and 90s that Ozempic has today and was even FDA-approved—but it was later discovered that the drug could cause irreparable and even fatal heart conditions and was eventually taken off the market in 1997. Mediator, a weight loss pill manufactured by Servier, one of France’s largest pharmaceutical labs, is linked to thousands of deaths. The pill, which was released in 1976, was prescribed for 33 years before finally being banned in 2009.11
Phentermine, which makes up the “phen” in fen-phen and is similar in structure to amphetamines, was FDA-approved in 2011 and is still prescribed today for weight loss. Serious side effects of the drug include pulmonary hypertension (high blood pressure in the lungs) and heart valve disease.12
Some other weight loss drugs that picked up steam over the past century include HCG, Obetrol, orlistat (sold under brand name Xenical), and lorcaserin (sold under brand name Belviq).
The long-term effects of semaglutide are still unknown but, based on the notorious history of weight loss drugs, warrant extreme caution.
If you would like to receive an e-mail notice of the most recent articles published in The Vaccine Reaction each week, click here.
Click here to view References:1 Skydsgaard N. Novo Nordisk says EMA raised safety signal on drugs including semaglutide. Reuters, June 22, 2023.
2 Bezin J, Gouverneur A, Pénichon M, Mathieu C, Garrel R, Hillaire-Buys D, Pariente A, Faillie JL. GLP-1 Receptor Agonists and the Risk of Thyroid Cancer. Diabetes care, 46(2), 384–390.
3 Lovelace B et al. People with diabetes struggle to find Ozempic as it soars in popularity as a weight loss aid. NBC News Jan. 12, 2023.
4 Aleccia J. FDA Warns People Not to Use Off-Brand Versions of Ozempic and Wegovy. Time Magazine, June 2, 2023.
5 Korte C. Ozempic side effects could lead to hospitalization — and doctors warn that long-term impacts remain unknown. CBS News June 12, 2023.
6 Gilbert D. Pfizer picks its pill in race for Ozempic-like weight-loss drug. Washington Post June 26, 2023.
7 Erman M, Mishra M. Pfizer scraps one weight loss drug in race to develop Ozempic rival. Reuters June 26, 2023.
8 Liu A. Novo Nordisk, AstraZeneca and Lilly will top pharma’s revenue growth in 2023: report. Fierce Pharma Jan. 6, 2023.
9 Bendix A. Ozempic and Wegovy force weight-loss companies to upend their longstanding strategies. NBC News June 4, 2023.
10 Janowiak J. “Unintended Consequences: A Case Study of Elvis Presley.” Journal of Health Education 30.6 (Nov./Dec. 1999): 364–66.
11 Guy J et al. Trial over weight loss drug linked to thousands of deaths starts in France. CNN Health Sept. 23, 2019.
12 Neal T. 9 Phentermine Side Effects You Should Know About. GoodRX Health Oct. 20, 2022.













2 Responses
Wish they had been as quick off the mark in assessing the Covid jabs.
I am constatly amazed after everything we have seen for years of pharmaceuticals being pulled out of the market that anyone would willfully take anything off label and for something like weight loss. I see posts avery day on Facebook about women inquiring about where they can buy Ozempic for wieght loss. So many people are so brainwashed about miracle cure that it is not surprising that pharmaceutical companies keep thriving.