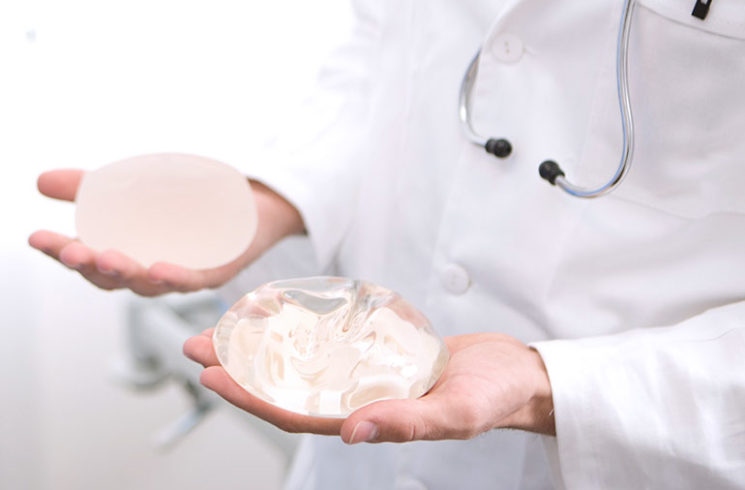
Breast implant surgery is the most common cosmetic surgical procedure in the United States, with more than 313,000 surgeries performed in 2018—a 48 percent increase since 2000.1 Silicone implants are the most common reconstructive breast surgery. In 2018, the American Society of Plastic Surgeons (ASPS) reported 101,657 breast reconstruction procedures, of which 78,814 used silicone implants.2
The U.S. Food and Drug Administration’s (FDA) Medical Device Reporting (MDR) is a post-market surveillance tool that the agency uses to monitor the performance of devices and detect any device-related safety issues, which contributes to benefit-risk assessments of medical device products.3
The FDA recently complied a report gathering data from the MDR database for all reports posted between Jan. 1, 2008 and Apr. 1, 2022 referring to adverse reactions to a saline or silicone filled breast prosthesis.4 The most common symptoms reported were fatigue (43.6 percent), joint pain (29 percent), brain fog (23.6 percent), anxiety (22.7 percent), hair loss (20.3 percent), depression (17.2 percent), autoimmune diseases (16.6 percent), rash (15.6 percent), headache (15.3 percent) and inflammation (14.7 percent).5
Breast Implant Illness Not Recognized As a Medical Diagnosis
Many patients and medical professionals use the term “breast implant illness (also known as “BII”) to describe the range of symptoms that some women report following breast implant surgery. The manifestation of BII symptoms has been reported with all types of breast implants regardless of the type of implant. Onset of reported BII symptoms ranges anywhere from immediately after surgery to years later.
Despite BII symptoms being very common among women with breast implants, breast implant illness is not recognized as a formal medical diagnosis and there are no specific tests or recognized criteria for medical professionals to make an official diagnosis.6
FDA Adds Black Box Warning to Breast Implants
In an 2019 FDA hearing, women with breast implants testified that their doctors did not warn them about the potential complications of breast implants. After the hearing, the FDA decided to restrict the sale of breast implants only to health care providers who offer patients a standardized checklist that explains risks of breast implants. The FDA now requires doctors to discuss with patients the risks involved and to give the patient an opportunity to sign off on the checklist, confirming they were informed about the risks.7
Jessica Everett, a businesswoman based in Houston, Texas who has breast implants, said:
The takeaway message is that it is truly dangerous for us getting the implants because we don’t know how they’re going to affect us. For some women, they don’t affect them—my mother has implants, and they have not affected her—but they have affected me. The medical community just needs to be educated on this in order to help other women with diagnosis and prevention.8
The FDA’s new warning states that all approved breast implants undergo safety testing prior to approval but, even so, there are risks associated with all breast implants. Some of these risks include additional surgeries, lymphoma, breast implant illness, scar tissue that squeezes the implant, breast pain, rupture of implants and infection.9
If you would like to receive an e-mail notice of the most recent articles published in The Vaccine Reaction each week, click here.
Click here to view References:1 Jalalabadi F et al. Breast Implant Utilization Trends in USA versus Europe and the Impact of BIA-ALCL Publications. Plastic and Reconstructive Surgery – Global Open 2021; 9(3).
2 Ibid.
3 U.S. Food and Drug Administration. Medical Device Reporting (MDR): How to Report Medical Device Problems. Feb. 18, 2022.
4 Ibid.
5 Ibid.
6 Ibid.
7 Christensen J. FDA adds boxed warning to breast implants. CNN Oct. 27, 2021.
8 Howard J. Women raise concerns about breast implant safety. CNN Mar. 26, 2019.
9 FDA. What to Know About Breast Implants. Nov. 16, 2021.
10 Dyer













6 Responses
I got saline implants because I was too young for silicone. I was just 19. I had extreme fatigue, brain fog, hair loss and shortness of breath. It was so bad that I paid a doctor out of pocket to remove them in his office because I couldn’t afford a hospital trip and my insurance wouldn’t cover removal. I instantly felt much better but I am convinced that these symptoms have not fully disappeared even after removal. I worry now about breast cancer since there has been so much damage to the tissue. Biggest regret and it’s such a shame that my own mother and grandmother made me feel as though I needed them in the first place, I was too young to think for myself.
God bless you dear. That’s heavy to carry without the full armor of GOD
I had a friend who is now in her early 70s. She had breast implants and the solution slowly leaked out over a number of years. She then developed a rare form of breast cancer–twice. Finally, she decides to get the implants taken out. Took a very long time to find a doctor who would do it–not sure why. Her main concern, over all this misery, was that her body would not look normal without the implants, as they were done after radical surgery. How sad to realize we put such pressure on women to do these things. Mammary glands are feeding units for newborns. They are a wonder of nature to nurture a child. That’s it in my mind.
To A. Vell,
Pressure from family is one of life’s strongest influences.
My family often steered me in the wrong direction, mostly due to their own insecurities.
That you were able to think for yourself through the implant poisoning and family dynamic is nothing short of a miracle.
I had a double mastectomy for breast cancer; tests showed it wouldn’t respond to chemo and that I had a 3% recurrance factor, covid shut down radiation. I had refused breast implants from the very beginning….now like Kathy Bates, the actress, I just go flat…best choice I every made…never wear a bra again…so freeing…2 1/2 years cancer free….
To A Vell ..
So glad to hear you had the implants removed. Do your research as the body knows how to heal itself if given what it needs! Anyone with implants will develop a capsule around them as the body recognizes the implants as a foreign object – capsule forms to try and ‘protect’ the body from the implants.
Where many surgeons do .. still many that do not (or choose to look the other way) is to remove the capsule when the implants are removed. The capsule is full of toxins and this can be removed even after explant surgery. If know the capsule was not removed or are not sure .. please do the research and find a surgeon who is experienced at removing breast implants and is knowledgeable of capsule removal as well. There is a website wwwbreastimplantsillness.com that has loads of information.
Prayers to you on your healing journey.