The European Medicines Agency (EMA) announced on Aug. 11, 2021 that it is investigating a possible link between the experimental messenger RNA (mRNA) biologics developed by Pfizer/BioNTech and Moderna/NIAID and an allergic skin condition called erythema multiforme, as well as two kidney disorders known as glomerulonephritis and nephrotic syndrome.1 2
Symptoms of erythema multiforme are the appearance of “symmetrical, red, raised skin areas” all over the body. The allergic reaction can cause itching, cold sores, fatigue, joint pain and fever.3
According to a paper describing the reaction and summarizing treatment options published in American Family Physician:
Erythema multiforme s an immune-mediated reaction that involves the skin and sometimes the mucosa. Classically described as target-like, the erythema multiforme lesions can be isolated, recurrent, or persistent.4
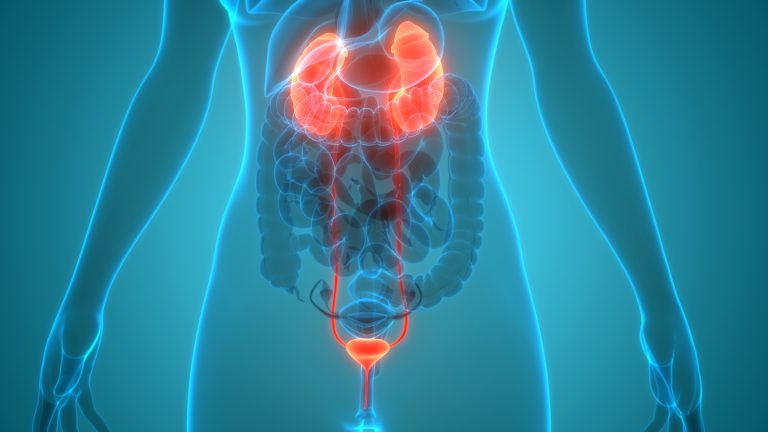
Glomerulonephritis (GN) is characterized by inflammation of the “tiny filters” (glomeruli) in the kidneys that remove “excess fluid, electrolytes and waste” from the bloodstream and pass them into the urine. Severe or chronic glomerulonephritis can damage the kidneys.5
According to Cleveland Clinic, GN can develop secondary to endocarditis (infection of heart valves), other viral infections, diseases that inflame the blood vessels, and when the immune system attacks healthy parts of the body, such as with lupus, an autoimmune disease.6
Nephrotic syndrome (NS) occurs when there is damage to the “clusters of small blood vessels” in the kidneys that “filter waste and excess water” from the blood. The condition causes swelling, notably in the feet and ankles, and it can increase the risk of infections and blood clots.7
A recent study published in the Journal of the American Society of Nephrology evaluated kidney, cardiovascular, and mortality outcomes in adults with primary nephrotic syndrome and found that they were at increased risk for end stage kidney disease (ESKD), acute coronary syndrome, heart failure, ischemic stroke, venous thromboembolism and death.8
The EMA did not say how many people may have suffered from these two reported adverse events involving an allergic skin reactions or kidney disease following reciept of Pfizer/BioNTech’s BNT162b2, which is also known as “Comirnaty”9 and Moderna/NIAID’s mRNA-1273, also known as “Spikevax.”10 The European health agency confirmed that 330 million doses of Pfizer’s BNT162b2 and 43.5 million doses of Moderna’s mRNA-1273 have been administered in the European Economic Area (EEA) as of July 29.1 2
Other Safety Problems for COVID Shots in Europe and the U.S.
There have also been safety concerns in Europe with regard to the experimental AZD1222 and Ad26.COV2.S adenovirus vaccines developed by AstraZeneca/Oxford University and Johnson & Johnson/Janssen respectively. The concerns involve reports of blood clots in people who received these vaccines. On Apr. 14, the European Commission (EC) decided not to renew production and delivery contracts for Astra Zeneca’s AZD1222 and J&J’s Ad26.COV2.S.2 7
In June, the U.S. Food and Drug Administration (FDA) added warning labels to Pfizer’s BNT162b2 and Moderna’s mRNA-1273 after determining a “likely link” between these pharmaceutical products and cases of heart inflammation (myocarditis) in the United States.11 In July, the FDA added a warning label to J&J’s Ad26.COV2.S linking the vaccine to cases of a paralyzing neurological disorder known as Guillain-Barré syndrome (GBS).12
The EMA recently recommended that immune thrombocytopenia (low blood platelets leading to internal bleeding), dizziness, and tinnitus (ringing in the ear) be added as potential side effects to the label of J&J’s Ad26.COV2.S and GBS be added as a possible reaction to AstraZeneca’s AZD1222.13
If you would like to receive an e-mail notice of the most recent articles published in The Vaccine Reaction each week, click here.
Click here to view References:1 Aripaka P. EU regulator looking at new possible side-effects of mRNA COVID-19 shots. Reuters Aug. 11, 2021.
2 Dunleavy K. Pharma Pfizer, Moderna COVID vaccines face new safety probe in Europe over possible link to skin condition, 2 kidney disorders. Fierce Pharma Aug. 11,2 2021.
3 Johns Hopkins Medicine. Erythema Multiforme.
4 Trayes KP, Love G, Studdiford TS. Erythema Multiforme: Recognition and Management. American Family Physician 2019; 100(2): 82-88.
5 Mayo Clinic. Glomerulonephritis.
6 Cleveland Clinic. Glomerubnephritis (GN). Nov. 4, 2020.
7 Kansteiner F. Europe won’t renew AstraZeneca, Johnson & Johnson COVID-19 vaccine contracts next year: report. Fierce Pharma Apr. 14, 2021.
8 Go AS, Tan TC, Chertow GM et al. Primary Nephrotic Syndrome and Risks of ESKD, Cardiovascular Events and Death: the Kaiser Permanente Nephrotic Syndrome Study. J Am Soc Nephrol 2021; 32(8).
9 Bulik BS. The inside story behind Pfizer and BioNTech’s new vaccine brand name, Comirnaty. Fierce Pharma Dec. 23, 2020.
10 Bulik BS. Moderna locks up Spikevax name in Europe, joins Pfizer Comrinaty in wait for official brand approval in U.S. Fierce Pharma June 30, 2021.
11 Dunleavy K. Pfizer, Moderna COVID-19 vaccines will get label warning over rare heart risk, FDA says. Fierce Pharma June 24, 2021.
12 Weixel N. FDA adds new warning to J&J COVID-19 vaccine. The Hill July 12, 2021.
13 Aripaka P. No link found so far between menstrual disorders and COVID-19 vaccines, EU says. Reuters Aug. 6, 2021.












15 Responses
Just imagine how great it would be if the unvaccinated can get these cooties the vaccinated are shedding.
Just imagine how great it would be if the unvaccinated can get these cooties the vaccinated are shedding. Would it be possible to also have these maladies attack us if we are near the recently vaccinated/ I sure hope not!
I doubt it, if you have a healthy innate immune system, and try to keep away from the vaxxed as much as possible. The cooties you describe are the S proteins, not the mRNA packages. On inhalation they will meet the mucosal immune system and will be rapidly identified and cleared – professional antigen presenting cells such as macrophages are well able to clear non-self or deformed extracellular proteins. They digest the protein and express it on MHC (Major Histocompatibility Complex) class II, thus informing T-cells of the pathogens presence. This is why healthy kids don’t get symptoms (or antibodies) from covid, as their innate immune systems clear it immediately. Unfortunately for the vaxinated, the trillions upon trillions of prion proteins expressed by the mRNA completely bypass this wonderful system and end up clogging capillaries at best, arteries at worst.
They knew about shedding back in 2015 – go to flemingmethod.com under the EVENT 2021 tab
Yep, and that’s exactly how I got the ‘flu’ the last time I did quite a few years ago. The only person I had seen for days was a man who approached me to ask me a lot of questions, who then mentioned he had a ‘flu’ vaccine that day. That is the ONLY time I have had ‘flu’ since I decided to give away pharma anything and doctors many years before that. These injections will also have transmission of other kinds, but one that people really won’t realize is that many of those who have had the REAL jabs this time around will likely be emitting rogue EMF signals now, and so move away if you feel nauseous or start to get a headache around people. This affects the energy body, so learning to protect that part of ourselves isn’t a bad idea either, IMO.
This happened, later in the night, to my 65 year old un\/’ed mother after having a 30 min meeting in a small room with 3 \/’ed people. imho, im convinced it was shedding/transmission.
I’ve not taken the recommendations because I don’t trust the people pushing it.
Most of my family did and I went to a party with them before leaving hugs were given.
6 days later I had these legions the article is speaking about and haven’t had a normal bowel movement in a week. I firmly believe it was from shedding and now I’ll be keeping my distance.
I noticed it only after my husband got his second Astra Zeneca shot. I definitely felt off, and found that spraying the house, and bedsheets every day with vinegar and water (2:3)was hugely helpful. Luckily this only lasted about one month so I imagine J an J could be the same. With the mRNA’s of course it could be a lot longer or indefinitely – who knows..
I also changed our towels every day, and as well as a morning shower, had a quick rinse at night. Worth the trouble.
My husband just got the vaccine 2 weeks ago, he eas perfect ans started 5 days ago with a Critical imfection in his eyes terrible red, itchin, hurting he said it felt like having cristals in historia eyes, algo stomach aches a and dizZINESS that wont go sea, he got astra zenwca
My 48 y/o healthy husband with no chronic medical conditions was in for routine blood work in April 2021 and was found to have elevated kidney markers (was actually stage 3 kidney disease with NO PRIOR kidney problems his entire life). Then end of May broke out in a head to toe terribly itchy rash for 7 weeks. He was on max doses of oral steroids, quadruple antihistamines, and steroid creams for this awful rash for over a month. He got the vaccine Dec 2020 as one of the first rounds of vaccinated individuals, as he is a frontline physician working in the hospitals.
It will be interesting to see if others develop these problems months after having the vaccine.
He didn’t know who to report it to or even if it was vaccine related, so the manufacturers are clearly not tracking this if it is vaccine related.
Now what??!!! The FDA just added more warning labels of side effects from mRNA covid biologics. They literally are doing experiments on them when they take covid jabs.
I don’t know if there is a connection or not but I was vaccinated with the Moderna vaccine back in Feb. 2021. I had this break out of severe dermatitis all over my body and I’m still dealing with it. Now I’ve been having pain in my neck, they did an MRI and found inflammation at C1 C2 of my spine been in severe pain going on 12 weeks now. The only relief was when I went to Urgent care and they gave me a high dose of prednisone for 5 days! I was able to function. Today they did a blood test, don’t know the results yet. And meeting with a Rheumatologist in 3 weeks! This waiting is the worse and I live in a large city! Anyway, that’s my story. I’m regretting getting the covid vaccine. As I already have an auto-immune disease UC since ’97.
Three months after receiving my second mRNA vaccine my kidneys function went into a fee fall. It went from 40 to 18. I was sent to the hospital to start some aggressive treatment to stabilize the kidneys. I ask my kidney doctor if the vaccine is somehow responsible for what happened to me. He said it’s possible but they don’t know enough to say yes or no. For me it’s too much of a coincidence and believe it was the vaccine. I also had the blisters/sores in my leg mentioned above. I will be looking more into this.
All I see here is FUD (fear, uncertainty, and doubt)! I know the truth and here it is: you are a living, organic being in a living, organic environment teeming with with things that want to eat you, infect you, use you, and trick you. Welcome to life. Does life cause death, the answer is a resounding YES! Get over it, move on with life. Trust me, you’ll never get out alive!
Oh, and BTW, the Pfizer BioNTech COVID-19 mRNA vaccine was just approved today by the FDA. I got mine. Happy I was free to make a vaccine choice. Thanks TVR for fighting to allow me to choose.
OfcTrsh… the fda approved vaccine is not the one being given. The approved version is not even in production yet. So the doubt is for good reason.