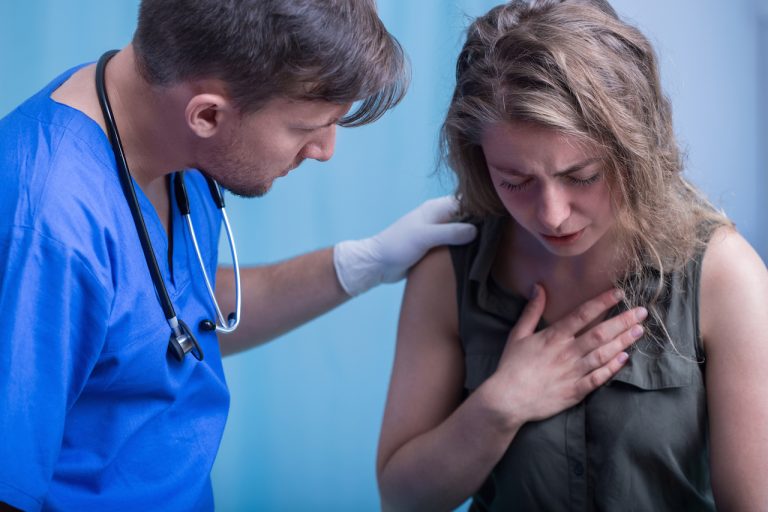
On Dec. 22, 2020, a patient at Decatur Morgan Hospital in Decatur, Alabama suffered a severe allergic reaction called anaphylaxis just minutes after receiving Pfizer/BioNTech’s experimental messenger RNA (mRNA) BNT162b2 vaccine for COVID-19.1 2 3
In a press release, the Alabama Department of Public Health (ADPH) stated the patient, who reportedly had a history of allergic reactions, was “immediately treated with anaphylaxis protocol at Decatur Morgan Hospital where the vaccine was administered” and was in stable condition and appeared to be recovering. “As required by the emergency use authorization (EUA) the adverse reaction has been documented and reported to the manufacturer,” the ADPH confirmed.1 2 3
According to Karen Landers, MD of the ADPH, examples of anaphylaxis symptoms, which can be life-threatening, are tightness in the chest, coughing, wheezing, swelling of the lips and tightness in the throat.4 5 The National Vaccine Information Center (NVIC) notes that other symptoms of anaphylaxis may include hives, hypotension (low blood pressure) and shock.6
Multiple Cases of Anaphylaxis After COVID-19 Vaccination Reported in U.S.
There have been multiple reports of anaphylaxis in following administration of the first of two scheduled doses of the BNT162b2 vaccine during the past month and the U.S. Food and Drug Administration (FDA) is investigating these cases to determine the cause of the severe allergic reactions.7 8 9 10
Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, has suggested the “culprit” behind the reactions could be a chemical ingredient called polyethylene glycol (PEG)—a polymer derived from petroleum used as an excipient (a diluting agent) in both the BNT162b2 vaccine and Moderna’s COVID-19 vaccine known as mRNA-1273.7 11 12
On Dec. 23, another case of anaphylaxis following vaccination with the BNT162b2 vaccine was reported in New York City. The case involved a health care worker who suffered a “significant allergic reaction.” The individual, who works for the Mount Sinai Health System, was reported to be in stable condition and recovering.13 14 15 16
Click here to view References:
1 Mahan A.First reported severe allergic reaction following COVID vaccine happens in Decatur. WAFF 48 Dec. 23, 2020.
2 Rayburn J, Borrelli S. Person Suffers Severe Allergic Reaction After Getting Coronavirus Vaccine at Decatur Morgan Hospital. WAAY 31 Dec. 23, 2020.
3 Whites-Koditschek S. Alabama patient recovering after allergic reaction to COVID vaccine. AL.com Dec. 23, 2020.
4 Fambro C. ADPH explains COVID-19 vaccine allergy is rare. 6 WBRC Dec. 23, 2020.
5 Balentine JR. Severe Allergic Reaction (Anaphylactic Shock). eMedicineHealth Aug. 20, 2020.
6 What is Polio Vaccine? National Vaccine Information Center.
7 Axelrod T. FDA investigating allergic reactions to Pfizer vaccine reported in multiple states. The Hill Dec. 19, 2020.
8 Fisher BL. Over 3,000 “Health Impact Events” After COVID-19 mRNA Vaccinations. The Vaccine Reaction Dec. 22, 2020.
9 Sun-Times Wire. Vaccinations resume at Condell Medical Center after pause following negative reactions. Chicago Sun Times Dec. 19, 2020.
10 WGN Web Desk. Advocate Condell Medical Center temporarily pauses vaccinations after 4 employees experience reactions. WGN 9 Dec. 19, 2020.
11 Polyethylene glycol. Wikipedia.
12 Moore S. What is Polyethylene Glycol (PEG)? News-Medical.Net.
13 Eyewitness News. NYC health care worker suffers ‘significant allergic reaction’ to COVID vaccine. Eyewitness News 7 Dec. 23, 2020.
14 Hicks N, Steinbuch Y. NYC health care worker has city’s first allergic reaction to COVID-19 vaccine. New York Post Dec. 23, 2020.
15 Honan K, Vielkind J. New York City Health-Care Worker Has Severe Adverse Reaction to Covid-19 Vaccine. The Wall Street Journal Dec. 23, 2020.
16 Williams J. New York health care worker experiences serious adverse reaction to coronavirus vaccine. The Hill Dec. 23, 2020.











10 Responses
Thanks for your work, what you do is greatly appreciated. So i have noticed drive through shot clinics on the news, is that actually how this vaccine will be given? If so are the car insurance company’s on board with the payouts should a recipient have a reaction and cause a traffic accident while driving away? Just wondering. ?♀️
These people were in or near a facility that provided epinephrine and airway assistance. What about those in a nursing home who do not have standing orders for epinephrine? This will not go well.
Does anyone see any hope for BigPharma to stop their vaccines?
So what do we think will happen when CVS starts giving millions of these? Do we think they’ll be standing by with an EPI-PEN and what if that doesn’t work?
Might PEG be an adjuvant used to rouse up the recipient’s immune system, rather than a ‘diluting agent’ as the FDA claims? What if previous exposure to PEG, as reported by ChildrensHealthDefense.org , has already primed the immune system to react, strongly (to the PEG)? btw, does this have any relevance for women with breast implants??
still trying to find source of Mrna . I think it’s from fetal stem cells . They are saying it’s manufactured Mrna . But I believe they make the 1 strand that that stimulates the cell to fight covid , insert it into fetal Mrna and call that ( misleadingly) manufactured Mrna
This happened to a couple of nurses in the UK before vaccination started in the US. As a result, they stated the vaccine should not be given to people with a history of severe allergic reactions, such as this patient, so WHY was it given to her? They have also said everyone being given the vaccine should be observed for at least 15 minutes before being allowed to leave. Again, not happening in the US where there is a slap dash attitude towards giving this vaccine.
Texas has decided to ignore CDC advice and give it to pregnant women!
There have been a total of 6 anaphylactic reactions in the US. I think it would be more helpful to readers of you stated that instead of just “multiple reports.” “Multiple” sounds scary. 6 is a very small reasonable number, out of the 80,000+ who have received the vaccine. Are you trying to scare, or are you trying to provide factual information?
Anaphylactic reactions are possible after ANY medication is given.
Why would any rational person take an experimental vaccine given Emergency Use Authorization when a safe and effectuve therapy exists using calcifidiol, an analog of vitamin D.
See https://pubmed.ncbi.nlm.nih.gov/32871238/
Maybe when information is controlled rationality is suspended,
Please have a look http://bit.ly/vaxxmap (Covid-19 Vaccine Side Effects World Map)