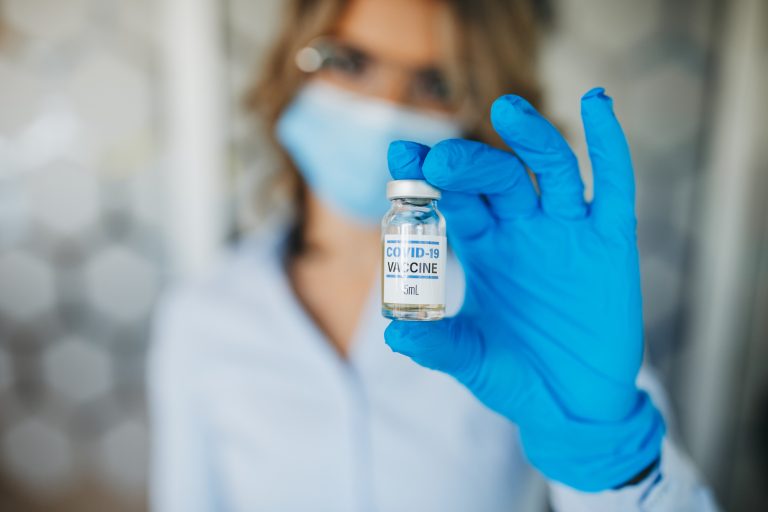
The German biotech firm BioNTech SE issued a joint press release with its American partner Pfizer, Inc. on Nov. 9, 2020 to announce that interim analysis of a global Phase 3 study of their mRNA-based SARS-CoV-2 vaccine appeared to demonstrate an efficacy rate “above 90%”measured seven days after the second dose.1
“This is really a spectacular number,” said Akiko Iwasaki, MD, an immunologist at Yale University. “I wasn’t expecting it to be this high. I was preparing myself for something like 55 percent.”2
Pfizer’s press statement, which was highly publicized and sent hopes and global stock markets surging, was based on 94 confirmed cases of COVID-19 in a trial that has enrolled nearly 44,000 people. Almost 39,000 of the participants have received both doses of the experimental messenger RNA (mRNA) vaccine known as BNT162b2.3 Pfizer and BioNTech did not release any vaccine safety data.
Experts Urge Caution as Full Data Set is Not Yet Available
Halfway through BioNTech’s announcement that a first peek at early data suggests its COVID-19 vaccine signals a “great day for science and humanity,” came this cautionary note: “As the study continues, the final vaccine efficacy percentage may vary.”
Eleanor Riley, PhD, professor of immunology and infectious disease at the University of Edinburgh, said:
At face value, this is exceptionally good news: a vaccine that is 90% effective at preventing symptomatic cases of Covid-19 and with millions of doses available by the end of the year. However, the full dataset on which the claim is based has not yet been released, and so we don’t know exactly what has been found.
Historically, important scientific announcements about vaccines are made through peer-reviewed medical research papers that have undergone extensive scrutiny about study design, results and assumptions, not through company press releases.4
Technology So New We Don’t Know What Questions to Ask
The Pfizer vaccine was designed with a brand-new type of vaccine technology that uses synthetic mRNA intended to instruct the body’s cellular machinery to make some of the SARS CoV-2 proteins, but not the entire SARS CoV-2 virus. When the vaccine is injected into the body, the immune system is expected to make antibodies against those parts of the virus. Basically, mRNA vaccines are intended to biohack—through genetic modification—a human being’s cells to produce protein parts of a virus.5 In effect, the cells of the body become a de facto vaccine manufacturer.
According to Amesh Adalja, a senior scholar at the Johns Hopkins University Center for Health Security and an infectious disease expert, an mRNA vaccine has never been approved to prevent any other disease, so this is a big first for the country.6
“This is great, this is American ingenuity,” said Matthew Heinz, MD, a hospitalist in Tucson, Arizona, who helped coordinate the country’s Ebola response under the Obama administration. “But we may not know what questions to even ask, just because this [technology] is very new.”7
Participants Evaluated for Illness Seven Days After Second Dose
In July, Pfizer and BioNTech initiated a Phase 3 trial of their coronavirus vaccine. Half of the people got the vaccine, while the other half got a placebo of salt water in two doses 21 days apart. The companies then waited for seven more days to see if people got sick to determine if the vaccine offered any protection.
So far, 94 participants out of nearly 44,000 have gotten sick with COVID-19. An independent board of experts looked at how many of the participants got the vaccine and how many got the placebo. The interim analysis suggests that very few people who were vaccinated got COVID-19.
The data was “blinded,” meaning that no one except the independent board—not the volunteers, doctors, or the company’s top executives—knew how many of the 94 people sickened by the virus got the vaccine or the placebo.8
The trial will continue until 164 cases of COVID-19 are diagnosed among the participants. At that point, the study will be complete and the results analyzed.
Significant Questions About Effectiveness Unanswered
Although the efficacy of the new vaccine (whether it “works” in a trial setting) may have been established by the Pfizer trial, details about the nature of the infections that vaccine can protect against, such as whether COVID-19 infections are mostly asymptomatic or mild cases and/or also include significant numbers of moderate and severe cases—are not yet available.
In an editorial published on October 22, 2020 in the British Medical Journal (BMJ)9 Peter Doshi, PhD, BMJ associate editor and professor of Pharmaceutical Health Services Research at the University of Maryland School of Pharmacy, noted that based on the published Pfizer trial protocol, people with only a cough and a positive laboratory test could qualify as meeting the definition of an “event.” He noted, “In the trials, final efficacy analyses are planned after just 150-160 “events, that is, a positive indication of symptomatic covid-19, regardless of severity of the illness.”
Duration of Vaccine-Induced Immunity is Unknown
The Pfizer announcement covers people who got two shots between July and October, but it doesn’t indicate how long protection will last or how often people might need boosters.
“To me, the main question is what about six months later, or even three months later,” says Rafi Ahmed, PhD, an immunologist at Emory University. And although little is known about the vaccine’s long-term effectiveness, that is unlikely to hold up its use, says Ahmed.10
“Generally, to ascertain how long protection lasts, follow up studies will be required to detect levels of both types of immune responses–antibody and T cell–as well any repeated exposure risks,” says Beate Kampmann, professor at London School of Hygiene and Tropical Medicine.11
Can Vaccinated Individuals Still Carry and Transmit the Infection?
It is unclear from the trial whether vaccinated people who show no symptoms or only very mild symptoms of COVID-19 can still be carriers of the virus and could spread the disease to others, even though they themselves presumably would be immune to its effects.12
Paul Hunter, professor of medicine at the University of East Anglia said:
It is not yet clear whether or not the vaccine could protect against coronavirus infection or simply against developing symptoms once you are infected. If it’s stopping infection then, by definition, it should be stopping transmission from one person to another. What we can say is that the vaccine stopped clinic symptomatic infection, but there is uncertainty over asymptomatic infection. But we will hopefully learn that as we go forward.13
Pharmaceutical companies argue it will be difficult to determine whether the Pfizer vaccine, or others in late stage trials, can achieve this because it would involve routinely testing trial participants. “You can’t do that with 45, 000 people,” says Kramer, because it involves swabbing people twice a week for very long periods, which results in five-to-ten times longer trial length and higher costs.14
In his BMJ editorial, Doshi pointed out, “None of the trials currently under way are designed to detect a reduction in any serious outcome such as hospital admissions, use of intensive care, or deaths. Nor are the vaccines being studied to determine whether they can interrupt transmission of the virus.”15
Are Trial Results Generalizable to People Who Will Get the Vaccine?
Pfizer did not disclose what percentage of its trial volunteers are in the groups most likely to be hospitalized or to die of COVID-19—including people over 65 and those with diabetes or obesity. This is a key point because many vaccines, particularly for influenza, may fail to protect the elderly though they protect younger people. “How representative are those 94 people of the overall population, especially those most at risk?” asked Walt Orenstein, MD, professor of medicine at Emory University and former director of the immunization program at the Centers for Disease Control and Prevention (CDC).16 Pfizer says it’s not yet evident whether the vaccine works on those who make up the worst COVID-19 cases—people who require hospitalization for the disease.”17
Children above the age of 12 and adults aged up to 85 are included in the trial but data broken down by age has not yet been released. Most vaccines do not work as well in older people as they do in younger people. This is not surprising as older people don’t always mount an effective immune response to a natural infection either.18 “We don’t know yet if it works in the population that needs it most, which is elderly,” says Florian Krammer, MD, a virologist at Icahn School of Medicine at Mount Sinai.
Both the National Academy of Medicine and the CDC have urged that older people be among the first groups to receive vaccines.
Does Benefit Outweigh Risk for Children?
In October, the FDA granted permission for Pfizer to enroll children as young as 12 years in their vaccine trials.
Cody Meissner, MD, a pediatric infectious disease vaccine expert at Tufts Children’s Hospital and a member of the Vaccines and Related Biological Products Advisory Committee (VRBPAC) of the US Food and Drug Administration (FDA), raised concerns about the safety of testing a COVID-19 vaccine on children, saying that since the virus’ effect on children is so “mild” public health officials had better be “very sure about safety of a vaccine.”
Meissner told the panel that most adverse reactions to vaccines come within six weeks of being immunized. “To me, we’ve got to be very sure that these vaccines do not elicit an adverse reaction that may be delayed,” he told his fellow committee members. “Relatively speaking, it’s a very mild disease and I think we have to be very sure about safety of a vaccine in children.”
Meissner also said that if he were part of the FDA, he would “certainly want to be very convinced about the safety of a vaccine before I approved its use in children,” adding that “the pattern of disease is very different in children, and lumping them in with adults would cause me some discomfort.”19
Could False Positives Skew Trial Results?
COVID-19 tests aren’t 100 percent accurate. A study of 90 patients who were tested with a COVID-19 test developed by Danaher‘s Cepheid unit, which is being used in Pfizer and BioNTech’s study, produced two false positive results.
Pfizer and BioNTech’s study requires trial participants to have at least one symptom within four days before or after the positive test to be counted as a positive COVID-19 case, but many of the symptoms, including fever, cough, sore throat, diarrhea, and others, are associated with other more benign diseases. With 21,000 volunteers in the study having received the vaccine, it’s easy to imagine that there might be a few participants who get a false positive test and also have one of those rather benign symptoms.20
No Long-Term Safety Data on New Technology
Though the short-term safety data on this mRNA vaccine is being positioned as providing robust and reassuring proof of efficacy, researchers will need to continue following the study participants to see if anyone experiences adverse effects six months or a year after being vaccinated.
Given this is a brand-new type of vaccine that uses mRNA technology that hasn’t been approved for use in humans before, “it is not impossible that someone could be allergic to a component of it,” says Paul Hunter, professor of medicine at the University of East Anglia. “There are some side-effects–like a sore arm or fever–that are quite common with vaccinations.”21
According to Orenstein, scientists will have to keep an eye out for rare effects such as immune enhancement, a severe illness brought on by a virus’s interaction with immune particles in some vaccinated persons.22
Adverse Reactions in Over Half of Clinical Trial Volunteers
So far Pfizer and BioNTech have reported no serious safety concerns from their vaccine. A senior Pfizer executive told the news outlet STAT that side effects from the company’s vaccine appear to be comparable to those of standard adult vaccines but worse than those of the company’s pneumonia vaccine, Prevnar, or typical flu shots.23
Published results24 of the Phase I/II trials conducted in May-June 2020 involving 45 healthy adults between the ages of 18 and 55 reported adverse reactions, including fever, sleep disturbances, and injection site pain, in over 50 percent of the participants. Two participants suffered “severe” reactions. Grade 3 or severe reactions are described by the U.S. Department of Health and Human Services (HHS) as, “severe or medically significant but not immediately life-threatening; hospitalization or prolongation of hospitalization indicated; disabling; limiting self care” such as “bathing, dressing and undressing, feeding self, using the toilet, taking medications.”
Detailed analysis of adverse reactions from the Phase 3 trial is not yet available.
“Real World” Challenges May Diminish Effectiveness
Although the interim data looks promising, the jump to mass vaccinations presents new hurdles, in particular for the Pfizer and BioNTech mRNA COVID-19 vaccine that must be stored and shipped at minus 70 degrees Celsius (-94°F).25
The Pfizer vaccine, unlike other COVID-19 vaccines in late-stage testing, must be kept supercooled in special sub zero freezers or on dry ice from the time it is produced until a few days before it is injected. Messenger RNA quickly self-destructs at higher temperatures.
Pfizer has devised an elaborate system to transport the vaccine by truck and specially designed cases to vaccination sites. Public health workers are being trained to handle the vaccine, but it’s unclear what will happen in case there is a failure to keep the vaccine at minus 70 degrees Celsius. Mishandling the vaccine along the way from factory to the person being vaccinated in a doctor’s office, pharmacy, big box store or other venue would render the Pfizer-BioNTech COVID-19 vaccine ineffective, which could result in people incorrectly thinking they were protected after getting vaccinated when they were, in fact, not protected…
Moreover, the Pfizer-BioNTech vaccine requires that two doses be given, ideally 21 days apart. If people do not stick to the timetable, it may affect the vaccine’s ability to prevent COVID-19 disease.
Pfizer Will Seek Emergency Use Authorization
According to FDA guidelines, Pfizer cannot file for an emergency use authorization to distribute the vaccine until half of the participants in their study have been observed for any safety issues for at least two months following receipt of their second dose. Pfizer expects to cross that time threshold in the third week of November.26
Pfizer Moves Ahead With Manufacturing
Should the Pfizer-BioNTech mRNA COVID-19 vaccine receive emergency authorization from the FDA, Pfizer’s chief executive has said that it could have 30 to 40 million doses of the vaccine before the end of the year, enough for 15 to 20 million people to get an initial shot and get a booster three weeks later. The companies have stated they could ramp up production to 1.3 billion doses a year.27
In July, Pfizer and BioNTech announced a $1.95 billion deal with the U.S. government for pre-purchase of 100 million doses of their vaccine, and the option to buy an additional 500 million doses.28
References:
1 “Pfizer and BioNTech Announce Vaccine Candidate Against COVID-19 Achieved Success in First Interim Analysis from Phase 3 Study. Investors Biontech Nov. 9, 2020.
2 Thomas K, Gelles D. Zimmer C. Pfizer’s Early Data Shows Vaccine Is More Than 90% Effective. The New York Times Nov. 9, 2020.
3 Kirkey S. Lost in the Pfizer press release that vaccine is 90% effective: ‘Percentage may vary’. National Post Nov. 10, 2020.
4 Allen A. Five Questions to Ask About Pfizer’s Covid-19 Vaccine. The New York Times Nov. 10, 2020.
5 Mayer A. Leading COVID Vaccine Candidates Plagued by Safety Concerns. The Defender Nov. 13, 2020.
6 Reis J. What Medical Experts Think About Pfizer’s New COVID-19 Vaccine. Huffpost Nov. 11, 2020.
7 Ibid.
8 Zimmer C, Thomas K. Pfizer’s Covid Vaccine: 11 Things You Need to Know. The New York Times Nov. 12, 2020.
9 Doshi P. Will covid-19 vaccines save lives? Current trials aren’t designed to tell us. British Medical Journal Oct. 22, 2020.
10 Callaway E. What Pfizer’s landmark COVID vaccine results mean for the pandemic. Nature Nov. 9, 2020.
11 Grover N. 6 key questions about the Pfizer/BioNtech Covid-19 vaccine. The Guardian Nov. 10, 2020.
12 Grothaus M. Pfizer’s COVID-19: 6 things to know. Fast Company Nov. 9, 2020.
13 See Footnote 11.
14 See Footnote 5.
15 See Footnote 9.
16 See Footnote 4.
17 See Footnote 12.
18 See Footnote 11.
19 Murdoch A. Infectious disease expert at FDA meeting raises concern about ‘safety’ of kids getting COVID vaccine. LifeSite News Oct. 29, 2020.
20 Orelli B. 3 Reasons Pfizer and BioNtech’s Coronavirus Vaccine Might Fail Even If IT Works. The Motley Fool Nov. 1, 2020.
21 See Footnote 11.
22 See Footnote 4.
23 Szabo L. Aleccia J. Covid-19 vaccines may have potentially unpleasant side effects. NBC News Nov. 12, 2020.
24 Mulligan MJ et al. Phase 1/2 Study to Describe the Safety and Immunogenicity of a COVID-19 RNA Vaccine Candidate (BNT162b1) in Adults 18 to 55 Years of Age: Interim Report. medRxXiv July 1, 2020.
25 Miller J. Shot in the dark: Early COVID-19 vaccine efficacy explained. Reuters Nov. 12, 2020.
26 Herper M. Covid-19 vaccine from Pfizer and BioNTech is strongly effective, early data from large trial indicate. STAT Nov. 9, 2020.
27 See Footnote 8.
28 Weise E. US cuts $1.95 billion deal with Pfizer for 100 million doses of COVID-19 vaccine. USA Today July 22, 2020.













24 Responses
I find no comfort that the results of only 164 out of 43,444 participants being tested ( 4ths of 1%) will be the determining factor for the country or the whole world as to how effective a vaccine might be. I do not believe in vaccine miracles, expect that they are created by pharma for monetary purposes, and to me this is far from anything viable. When no coronavirus vaccine has ever been created, and considering we are told this is the mother of all viruses (total nonsense), I have no faith in these results. They don’t matter to me because I will resit ALL vaccines until Big Pharma steps forward and decides to take full responsibility for the outcome of there usage. And just what is an RNA altering vaccine? Will we get the truth? NEVER! I would suggest that all employees at Pfizer and BioNTech be the first to try out this new miracle vaccine. I bet not one of them will ever take it.
This nurse, having worked 2 years recently in a clinic, witnessed how vaccines were transported, stocked and eventually injected sees first concern is keeping vaccine supercool. This would be difficult, at best, to maintain along the ‘manufacturer to consumer pathway’ without rendering it less than optimal. Second concern, does vaccine interfere with DNA, RNA gene replication within the body? As this is BRAND NEW, and a once trailed RNA vaccine failed miserably mid-1900’s, this needs fastidious follow up, for years, as we are talking about impacting future generations. Lastly, with sooo much yet to determine about COVID transmission, effectiveness and whether benefits outweigh risk before rolling out to general public we should Keep our COOL and TEST ON before producing mass quantities, please. We do have time to GET THIS ONE RIGHT, right???
Yes your genetic make up will be “corrupted”. They are going to take a sequence of your genetic makeup located on Chromosome 8. A retrovirus is actually dead, how it becomes alive is that you need a catalyst to INITIATE an autoimmune response. This is why you keep hearing the terms “safe and effective” …..NOT SAFE , BUT effective at causing your immune system to attack itself. Something has to be in a certain state of acidity ….something out of balance for that to REPLICATE. Whether it be inside of your body or environmental factors or both. So yes messenger RNA (mRNA) has NEVER BEEN injected into anyone since the history of immunizations. This is what I believed in Wuhah….they injected them some type of genetic material that would replicate at a rapid rate. Then in October they implemented 60gh millimeter wave length technology in October of 2019. Now combine that with elders who are already immunocompromised in poor health, eat no nutrient dense food, take synthetic Big Pharma meds, morbidly obese , exposure to electromagnetic radiation…..woooo weeeeeee. What do retroviruses love…Toxic Cells with a Toxic Cellular HOST!
Interesting angle on potential issue with this m RNA vaccine. Reactivation of any of the many viral genomes that are currently dormant in our cells. I am wondering if the mRNA keeps our cells churning out viral proteins and is so stimulating to the immune system that the immune system goes on overdrive and causes huge problems. I developed severe asthma after a viral infection. Virus was gone, but immune system didn’t calm done until a doctor finally gave me a huge shot of prednisone and a Medrol dose pack.
Bet there plenty of others like me.
good luck people,i’ll take my chances without this mystery drug,they do more testing on drugs for animals,which is what they think we are,sheep,can you all say bbaaaaa!!
There are multiples things very wrong with this. First and foremost is What did they use a placebo? Was it another vaccine like so many other times? Was it a totally benign substance? I do believe placebos used were a substance that would cause a double jeopardy nullifying any true meaningful results. Come on folks we know how these companies work. Wake up!
One CANNOT have a safe vaccine (if ever) when one is traveling at warp speed. Think about it. This is not a safe procedure nor vaccine.
In other words, we would be their science project for many years and we will find out what the effects are at the same time the experts do. In other words, how can this be preventive medicine practices. It is unbelievable what companies will do for a profit! There is no way I will become their Guinea pig!
It concerns me when “RNA-based” is used before the name of this vaccine. I saw a video a couple months ago by a doctor who was warning about a new vaccine coming out combined with RNA. She said that it could essentially change your DNA in time, if I understood correctly. Do I have a right to be concerned?
I saw a video with this same information and that RNA referred to stuck in my mind. It was not a positive message. The doctor did say that there could be a possibility of it changing our own DNA. No thanks!
Maybe Im missing something in the article. If a participant didnt test positive after receiving the vaccine they were deemed protected? How was efficacy determined exactly?
You can find their protocol online, efficacy 1- [rate of infection in vax group/ rate of infection in placebo group].
So if you have hypothetical participant groups of vax:placebo = 20,000:20,000
And you have hypothetical results:
7 in vax group get covid
And 87 in placebo get covid,
You get 1-[(7/20,000)/(87/20,000)]=0.92
So, you could call that “over 90% effective”. But this is BS because these trial participants are not all living in the same place with the same exact exposure levels, how would we ever know if the vaccine “protected” them, or they live in a rural area with minimal exposure?
I would certainly feel better if we hadn’t had such recent vaccine disasters. The Dengue vaccine – Sanofi Pasteur said they tested it on 30,000 people – great safety data. WHO mandated it for all kids 9-16 in Philippines. A disaster. Then there was the 2009 H1N1 “ pandemic “ vaccine. 1 out of every 16,000 developed narcolepsy/ cataplexy. Recently, polio vaccine caused polio outbreak in Africa.
So, how can we trust these vaccine manufacturers ?
Honestly, I am not concerned with the efficacy rate; if it helps even one person, that is wonderful. As the father of two vaccine-injured sons, however, I am most concerned with the SAFETY of this drug. This vaccine is using a brand new technology never before used on humans, and NO ONE knows what the short-term negative effects will be, never mind the long-term. In their haste to “cure” this intentionally-unleashed pandemic, the powers that be and their not-legally-liable Big Pharma friends have thrown virtually all safety protocols out the window. It is not unreasonable to say that every recipient of this and all of the soon-to-be released other Covid vaccines will be a walking guinea pig. That should scare the living daylights out of every thinking person.
Germ theory was disproven by Bechamp in the 19th century.
More vaccines please! LOL It’s time to thin the herd…
Dr.Frankenstein would be very proud.
Last Monday I spoke to the Pfizer clinical study department and the gentleman answered when I asked if they have done challenge tests he said no but they were ‘discussing’ it but couldn’t promise any would be done, animal or human!
He also said, in agreement to my comment on same, that “to date no mRNA vaccine has been recommended for human testing or use.
These criminals want to foist an unsafe, incompletely tested vaccine on millions of people. Is it greed alone or the Globalist depopulation agenda that drives their villainy?
Challenge trials on animals showed, lung necrosis and scarring, liver failure, venous and arterial scarring….because, it is theorized, that when a vaccinated individual is exposed to
the wild virus the body’s immune system goes on hyper drive attacking the systems that contain the virus DNA from the vaccines. It may take a bit of time for individuals to succumb to the damage wrecked in their bodies because of this vaccine…but hey the money will already be in the pockets of the perpetrators and with nothing to lose because they are protected by law from any recriminations or consequences, what do they care.
I for one WILL NOT submit my body to any vaccine no matter the consequences the state or federal governments will impose!
Yes, this is what happened with Sanofi Pasteur’s Dengue vaccine in the Philippines. 600 kids died, and exposure to the virus after vaccination may cause the hyper immune response.
My friend told me that Pfizer was manipulating statistics to get the 95% figures. How does this really add up?
Somebody will have to explain to me why it’s ethical to give placebos to patients in clinical trials, but Big Pharma has always said it would be “unethical” to conduct a definitive study of vaccinated versus unvaccinated populations.
Because they know it will show them up, that is all really…..it wont fit their BS narrative.
For everyone who talks about the lack of animal testing you need to understand that animal experimentation and testing cannot be extrapolated to humans because of differences in physiology, metabolism, bio chemistry, genetics and environment. It is impossible to know the damage that will be inflicted on humans by drugs and vaccines from animal tests. It is not science. It does not work. A long list of drugs tested on animals which then went on to harm and kill humans is available on our website. Furthermore, all vaccines contain decaying animal or insect matter and toxic chemicals that in and of themselves can cause serious diseases, paralysis and death. A religious belief in vaccines has been nurtured for decades in the population the same way belief in this phony “pandemic” was nurtured – fear mongering and pharma-owned media brainwashing.
“Cody Meissner, MD, a pediatric infectious disease vaccine expert at Tufts Children’s Hospital and a member of the Vaccines and Related Biological Products Advisory Committee (VRBPAC) of the US Food and Drug Administration (FDA): “Relatively speaking, it’s a very mild disease….” THIS SAYS IT ALL A VERY MILD DISEASE! ??????
Well, just doing the math using CDC’s own (week 46) data regarding testing vs symptoms… just over 2/10ths of a percent of those who tested positive for COVID actually sought any form of medical attention! Compare that statistic to the study’s targeted “164 out of 44,000” outcome, which is .37% (aka nearly 4/10ths of a percent) almost double! That’s about as simple as the argument gets!