A new study published in the journal Vaccine revealed there was a much higher risk of serious adverse events of special interest for randomized clinical trial participants who received a messenger RNA (mRNA) COVID-19 biologic when compared to participants, who received a placebo for both the Pfizer/BioNTech mRNA BNT162b2 (also known as “Comirnaty”) COVID shot and Moderna/NIAID mRNA-1273 (“Spikevax”) COVID shot.1
The serious adverse event risk for adult participants was higher than initially estimated at the time Pfizer and Moderna were granted an Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration (FDA) in December 2020 to distribute the COVID shots in the United States.
The study authors, who are affiliated with universities and research facilities in the U.S., Spain and Australia, examined available data from the Pfizer and Moderna phase III placebo controlled clinical trials on adults and found…
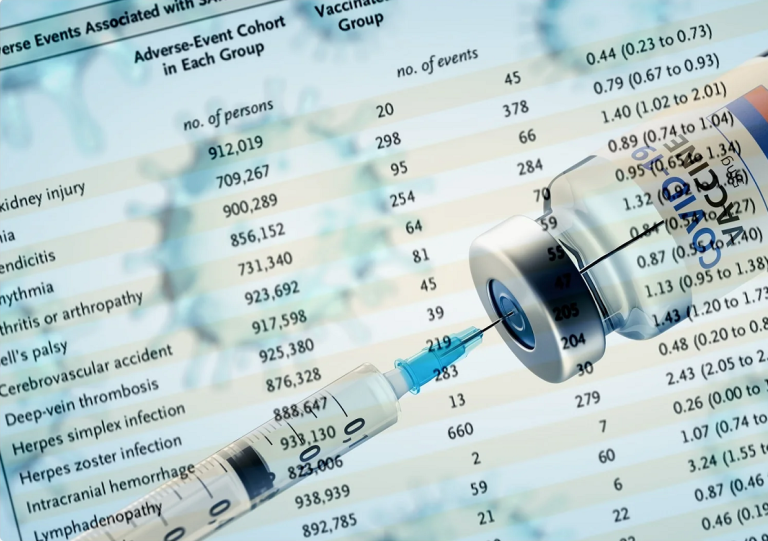
Using a prespecified list of AESI identified by the Brighton Collaboration, higher risk of serious AESI was observed in the mRNA COVID-19 vaccine group relative to placebo in both the Pfizer and Moderna adult phase III trials, with 10.1 (Pfizer) and 15.1 (Moderna) additional events for every 10,000 individuals vaccinated. Combined, there was a risk difference of 12.5 serious AESIs per 10,000 individuals vaccinated (95% CI 2.1 to 22.9). These results raise concerns that mRNA vaccines are associated with more harm than initially estimated at the time of emergency authorization.
The study results indicate that about 1 in 800 adults who received either the Pfizer or Moderna mRNA shots in the pre-EUA randomized clinical trials experienced a serious adverse event,
Excess Risk of Serious Adverse Events Exceeded Reductions in COVID Hospitalizations
When analyzing the Pfizer and Moderna COVID shot clinical trial data to determine harms versus benefit, the study authors found that the risk of trial participants suffering a vaccine adverse event exceeded reduction in COVID related hospitalizations. They said:
Rational policy formation should consider potential harms alongside potential benefits. To illustrate this need in the present context, we conducted a simple harm-benefit comparison using the trial data comparing excess risk of serious AESI against reductions in COVID-19 hospitalization. We found excess risk of serious AESIs to exceed the reduction in COVID-19 hospitalizations in both Pfizer and Moderna trials.
Brighton Collaboration Lists Potential Adverse Events of Special Interest for mRNA COVID Biologics
In 2020, the Brighton Collaboration, the Coalition for Epidemic Preparedness Innovations (CEPI) and Safety Platform for Emergency vACcines (SPEAC) developed a priority list of potential serious adverse events of special interest (AESI) relevant to COVID biologic clinical trials.2[ii]
On Mar. 5, 2020, the initial AESI list was approved based on the first scientific published studies of COVID shots from China. Thereafter, PubMed searches were conducted every day for new COVID clinical patterns and complications from COVID shots.3
From May to August 2020, Brighton Collaboration developed definitions for various COVID shot AESI. These definitions included Vaccine Associated Enhanced Disease (VAED), Acute Respiratory Distress Syndrome (ARDS), Multisystem Inflammatory Disease in Children (MISC), acute cardiovascular injury (including myocarditis, pericarditis, arrhythmia, heart failure, cardiogenic shock, stress cardiomyopathy, coronary artery disease and microangiopathy) and blood coagulation disorders (including thrombosis, thromboembolism and bleeding disorder).4
The study in Vaccine investigated the association between the two mRNA COVID shots the FDA approved for distribution under an EUA and serious adverse events identified by the Brighton Collaboration using data from the phase III randomized, placebo-controlled human clinical trials held before the EUA was granted to Pfizer and Moderna. The study investigated the clinical trial data compared to findings from post-EUA authorization observational safety data of the mRNA COVID shots. The investigators conducted a comparison of risks and benefits to demonstrate the need for formal risk-benefit analyses of the mRNA shots.5
Study Authors Call for Release ALL the COVID Shot Clinical Trial Data
Study authors expressed disappointment with lack of access to individual data for the Pfizer and Moderna clinical trial participants, which is needed to fill in knowledge gaps and answer outstanding questions about the potential harm versus the benefit of mRNA COVID shots. They stated:
The excess risk of serious adverse events found in our study points to the need for formal harm-benefit analyses, particularly those that are stratified according to risk of serious COVID-19 outcomes. These analyses will require public release of participant level datasets,” adding that, “Full transparency of the COVID-19 vaccine clinical trial data is needed to properly evaluate these questions. Unfortunately, as we approach 2 years after release of COVID-19 vaccines, participant level data remain inaccessible.
If you would like to receive an e-mail notice of the most recent articles published in The Vaccine Reaction each week, click here.
Click here to view References:1 Fraiman J et al. Serious adverse events of special interest following mRNA COVID-19 vaccination in randomized trials in adults. Vaccine August 2022; 40(40): 5798-5805.
2 Ibid.
3 Safety Platform for Emergency vACcines. SO2-D2.1.2 Priority List of COVID-19 Adverse events of special interest: Quarterly update December 2020. Brighton Collaboration Dec. 23, 2022.
4 Ibid.
5 Fraiman J et al. Serious adverse events of special interest following mRNA COVID-19 vaccination in randomized trials in adults. Vaccine August 2022; 40(40): 5798-5805.











8 Responses
A decade from now, we will still be bandying about the mRNA gene therapy injection stats and lies. The difference is that millions more will be dead or injured for life. And we will be on booster #37 for all those vac-tards that love their injections.
Well seen that you don’t understand the numbers!!! This is another piece of NONSENSE!!!
Based on their Alpha and CI, they could be exaggerating the effect by more than SIX TIMES, which changes the analysis completely. It is HIGHLY dubious to depend upon data with such a significant CI – 20 events in this case. The authors aren’t exactly well published with one exception, who is more interested in ethics than science! Most have only been involved in two anti vaccine papers, revealing their personal BIAS. Only hardened anti vaccine idiots, who lack the education and ability to think critically will pay any heed to such NONSENSE. In the future, it is those who believe this claptrap who will be the vac-tards…
This information needs to her given to all of our loved ones whether or not they have already received the mRNA vaccines. This information should be spread by all media (but it is not). CDC and FDA should be scraped and new agencies with Dr Malone, McCullough and others in charge of the new agencies.
Just like the dangers of lead, asbestos, DDT, tobacco, Vioxx, etc we’re suppressed and profits were made even after lawsuits,
we’ll look back years later and see the same with mRNA technology, but without lawsuits due to zero liability for pharmaceutical companies.
September 13, 2022 ‘Unethical’ and up to 98 Times Worse Than the Disease: Top Scientists Publish Paradigm-Shifting Study About COVID-19 Vaccines By Dr. Jennifer Margulis and Dr. Joe Wang
A team of nine experts from Harvard, Johns Hopkins, and other top universities has published paradigm-shifting research about the efficacy and safety of the COVID-19 vaccines and why mandating vaccines for college students is unethical.
https://www.globalresearch.ca/unethical-up-98-times-worse-than-disease-top-scientists-publish-paradigm-shifting-study-about-covid-19-vaccines/5793272
Bottom line: experimental Covid vaccines are very reactive without sufficient data in the long run. As far as I know of, it’s far better to get Covid infection than getting a vaccine. However, pre existing conditions such as diabetes pose more of a challenge in fighting off Covid viral infection. Benefits vs risks can be a tricky balance when it comes to making an informed decision. ?
Just do the research and be as fully informed as possible. That’s what National Vaccine Info center is all about, not pro or anti vaccination movement.
Has anyone heard the recent news about the WHO (World Health Organization) saying they think the covid pandemic is close to ending? I saw a quick clip on my morning news today. I also read a brief article online today saying Biden wants everyone to get the newest vaccine before Halloween this year. So why the rush if the WHO is really going to call an end to their pandemic pronouncement? Is it because the WHO will ruin the newest vaccine sales? And having read this article on the severe vaccine adverse effects, why would anyone want the new and unproven 8-mice tested vaccine in the first place? Since I am finding so many conflicting views on the Internet, anyone have info to share? Thanks.
Try real not rare and childen’s health defense links. WHO really doesn’t promote our health but promotes more diseases in order to gain a huge profit.