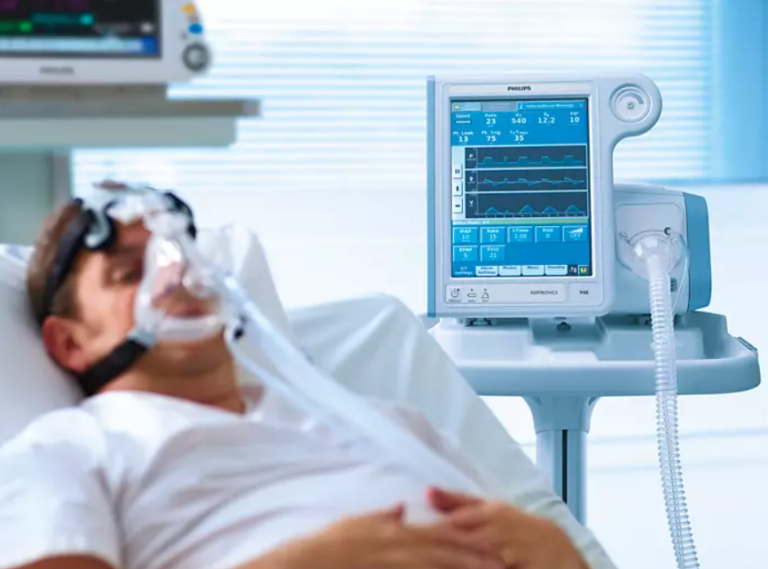
On Aug. 16, 2022 the U.S. Food and Drug Administration (FDA) issued an update on a June 2021 recall of certain ventilators, BiPAP (Bi-level Positive Airway Pressure), and CPAP (Continuous Positive Airway Pressure) breathing machines manufactured by Philips Respironics because of “potential health risks.”1 A list of the recalled devices made between 2009 and April 26, 2021 can be found here.
Polyester-based Polyurethane Can Degrade, Cause Injury
Philips Respironics recalled the devices because the polyester-based polyurethane (PE-PUR) sound abatement foam used to reduce sound and vibration could degrade. If this were to happen, black debris from the foam or certain invisible chemicals could be released into the device’s air pathway and inhaled or swallowed by the person using it.2 This could potentially end in in serious injury and require medical intervention to prevent permanent injury.3
The potential risks of inhaling or swallowing pieces of PE-PUR foam include irritation to the skin, eyes, nose and respiratory tract (airway), inflammation, headache, asthma and cancer-causing risks affecting organs such as kidneys and liver.4
The potential risks of inhaling chemicals released into the device’s air tubes from the PE-PUR foam include headache, dizziness, irritation in the eyes, nose, respiratory tract and skin, allergic reactions and another immune system reactions, nausea or vomiting, and cancer-causing effects.5
FDA Received 69,000 Complaints and 164 Death Reports Potentially Linked to Recalled Philips Breathing Devices
Following the FDA’s announcement in June 2021 recalling the affected Philips breathing devices, the agency received more than 69,000 complaints linked to the recall. Between April 2021 and April 2022, the FDA received 21,000 complaints and more than 120 reported deaths potentially related to the affected devices.6
Between May 1, 2022 and July 31, 2022, the FDA received many more medical device reports (MDRs)— over 48,000—involving Philips respiratory products, including 44 reports of death. MDRs include mandatory and voluntary reports of safety issues, injuries and deaths from patients, consumers and healthcare providers.7
France Launches Criminal Investigation into Philips Respiratory Devices Recall
France’s National Medicines Agency has received nearly 3,000 reports of adverse effects linked to Philips recalled ventilators and breathing machines. A criminal investigation into the company’s recall began on June 20, 2022. A spokesperson for the French prosecutors stated:
complaints filed on the grounds of aggravated deception, involuntary attacks on physical integrity, endangerment of the life of others and administration of harmful substances.8
If you would like to receive an e-mail notice of the most recent articles published in The Vaccine Reaction each week, click here.
Click here to view References:1 U.S. Food and Drug Administration. Update: Certain Phiips Respironics Ventilators, BiPAP Machines, and CPAP Machines Recalled Due to Potential Health Risks: FDA Safety Communication. Aug. 16, 2022.
2 Ibid.
3 FDA. Update: Certain Philips Respironics Ventilators, BiPAP Machines, and CPAP Machines Recalled Due to Potential Health Risks: FDA Safety Communication. Aug. 16, 2022.
4 Ibid.
5 Ibid.
6 Park A. One year into Philips’ ventilator foam recall, FDA tallies 124 deaths, 21,000 complaints. Fierce Biotech May. 23, 2022.
7 Park A. Philips recall sparks 48K complaints to FDA in 3 months, including 44 deaths. Fierce Biotech Aug. 17, 2022.
8 Park A. French prosecutors launch criminal investigation into Philips ventilator recall. Fierce Biotech Sept. 8, 2022.












10 Responses
I Have called numerous times reporting that I owned a Phillips Breathing Machine that was on recall. They always say “I’ll put your name on the list”. I have never heard from them. Not having a healthy machine to use it is affecting my health. What can I do to receive a replacement machine that is healthy to use????
You can register your machine on this site. Click on the brown button at the top: https://www.usa.philips.com/healthcare/e/sleep/communications/src-update. I can’t believe you weren’t told to do this when you called. This is how I got on the list. That said, it still took a year to get my new device. I had my old, old device that still worked, so I just switched to that one. You should write the company a letter and complain that you weren’t given correct information when you called.
I ended up paying out of pocket and renting a different machine at an online place because none of the local companies would help me other than “the list” you mentioned. Maddening.
does this include their nebulisers also does anyone know
We registered my husband’s VA-provided Phillips Respironics Dreamstation CPAP in June 2021, as the company promised a replacement for the models made with the deadly foam. The V.A. encouraged my husband to keep using it as the “benefits outweigh the risks”; a term which readers of this website are, I’m sure, more than familiar with. It was too scary to keep using it, so we paid cash for a brand new ResMed Air Sense 10.
It’s Sept 2022, and we haven’t received the replacement.
I use an A-Pap machine. never heard of a recall. should I worry? I was concerned about the plastic used (vinyls?) but nobody could tell me what the device is made of…
I have been on the list with my recalled machine for 15 months. Nothing new-they just say they are working on getting my new device by the end of the year. I went back to using my old machine. I think when I had to say that I used the SO Clean machine and my new machine went to the bottom of the list. Really frustrated!
How anyone could sleep with that large, ugly, electronic machine and the uncomfortable-looking contraptions attached to the face/head is beyond me. Better just let nature takes its course on its own and put up with any sleeping problems that one might have.
I waited a year to get my new machine. I was lucky because my old, old machine still worked, so I switched from the “new” machine to that one. Keep contacting them. I finally stopped calling the number they give you because they just kept putting me off and started making phone calls to other numbers I found on the website. I finally reached someone who helped me I think it was in Tech jSupport. Try this number: 1-800-345-6443. You need to make sure they have the information about your provider where you get your supplies and your doctor’s information. Verifying that information seemed to speed up the process. I got my machine within the month.
My wife and I both registered our units last year when the recall first came out. We found out by friends or ours who also used the same machines. If it weren’t for our friends letting us know I’m not sure when we would’ve found out about the recall. Phillips surely didn’t do much to let the public know about it. We eventually received our new units. I say new units! I have a feeling they’re refurbished units, but I hope the material they were supposed to change has really been done! I’m a little skeptical but we are having no adverse affects from using our machines.