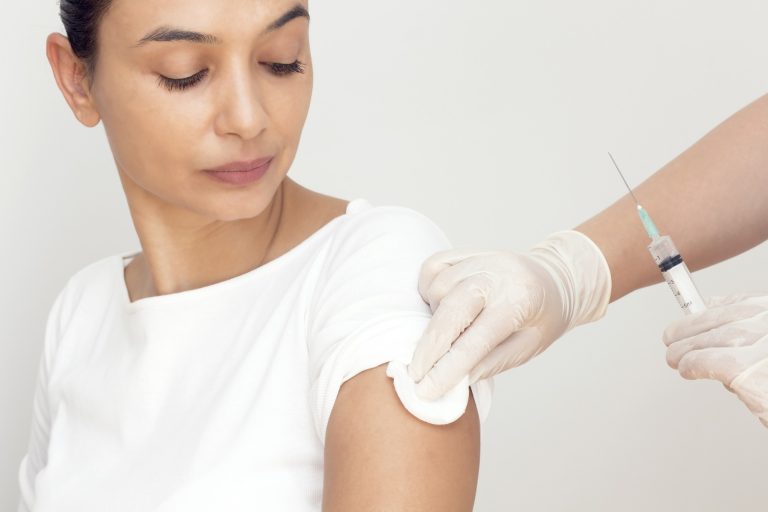
On Feb. 1, 2019, the Department for Public Health (DPH) in Kentucky announced it was investigating ” multiple infections associated with vaccinations” administered by a company called Location Vaccination of Mt. Sterling, Kentucky. The vaccinations were given at various business locations in Kentucky, Ohio and Indiana since Sept. 1, 2018.1
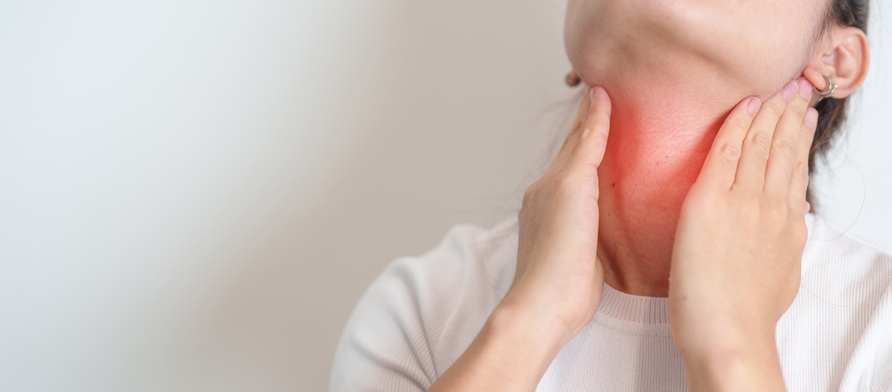
According a press release issued by the Commonwealth of Kentucky Cabinet for Health and Family Services, “Individuals with vaccination-associated infections related to this provider have experienced redness, pain or tenderness, swelling, and the development of hard lumps, or nodules, at the injection site.”1 It warns, “Symptoms may start from a few days to more than 12 weeks after vaccination. Medical care is advised since infections will likely not get better on their own.”1
DPH commissioner Jeff Howard, MD attributed the cause of the infections to “improper storage and handling” of the vaccine. “It is important that individuals vaccinated by this company seek appropriate medical evaluation and treatment,” Dr. Howard stressed.1 He did not specify which vaccine was responsible for the infections or vaccine(s) were thought to be responsible for the infections.
Severe local vaccine reactions, which produce varying degrees of redness, swelling, or pain at the injection site, are frequently reported and acknowledged reactions to vaccination.2 The CDC estimates that local reactions to childhood vaccines occur at the following rates and usually last from one to seven days:
- 1 in 4 children after diphtheria-tetanus-acellular pertussis (DTaP) shots
- 1 in 5 to 1 in 16 adolescents after Tdap booster shots
- 1 in 3 children after pneumococcal (PCV-13) shots
- 1 in 3 to 9 in 10 adolescents after HPV (Gardasil 9) shots
The World Health Organization (WHO) states that, “Local and systemic reactions such as pain or fever can occur as part of the immune response. In addition, other vaccine components (e.g. adjuvants, stabilizers, and preservatives) can trigger reactions.”3 Aluminum adjuvants in vaccines have been associated with severe local reactions, including large lumps at the site of the injection.
In 2014, vaccine researchers observed that not enough is known about the biological mechanisms and systemic toxic effects of local reactions:
Adjuvants are necessary components to warrant the efficacy of vaccines, however the overstimulation of the immune system is also associated with adverse effects. Local reactions are the most frequent manifestation of toxicity induced by adjuvanted vaccines and, with the exception of the acute phase response (APR), much less is known about the systemic reactions that follow vaccination.4
There have been reports in the medical literature of abscesses forming at the site of vaccine injections, both sterile abscesses5 and abscesses that involve a bacterial infection because a microbe (such as streptococcus or staphylococcus bacteria) contaminates the vaccine. Multi-dose vaccine vials can become contaminated from use of unsterile needles or pathogenic bacteria can enter the skin at the injection site if the person administering the vaccine does not follow skin disinfection protocols. Bacterial cellulitis (local sepsis) at vaccine injection sites has been reported and usually requires antibiotics.6
On Feb. 2, it was reported that the source of the vaccine “contamination” was traced to one Mt. Sterling doctor, who provided vaccines to businesses located in Alexandria, Butler, Georgetown, Lexington, Louisville, Paris, Maysville, Mt. Sterling and Winchester, and is no longer administering vaccines. The final words from DPH commissioner Howard to the public was framed as both a warning and an affirmation that more vaccinations are a good idea. He said, “If you received vaccine from this provider, we strongly encourage you to consider getting another round to ensure you are fully immunized and not at risk for contracting illness.”7
References:
1 Hogan D, Fisher, B. Department for Public Health Investigates Vaccination Associated Infections. Commonwealth of Kentucky Cabinet for Health and Family Services Feb. 1, 2019.
2 Centers for Disease Control and Prevention. Possible Side-effects from Vaccines. CDC.gov. July 12, 2018.
3 World Health Organization. Vaccine Safety Basics: Adverse events following immunization. VaccineSafetyTraining.org 2019.
4 Batista-Duharte A, Portuondo D et al. Systemic immunotoxicity reactions induced by adjuvanted vaccines. Int Immunopharmacology 2014; 20(1): 170-180.
5 Klein NP, Edwards KM et al. Recurrent sterile abscesses following aluminum adjuvant-containing vaccines. BMJ Case Rep Mar. 17, 2009.
6 Cook IF. Best vaccination practice and medically attended injection site events following deltoid intramuscular injections. Human Vaccines & Immunotherapeutics 2015; 11(5): 1184-1191.
7 WKYT. Health department says Kentucky-based vaccine provider caused infections. WSAZ NewsChannel 3 Feb. 2, 2019.











18 Responses
The DPH commishioner recommends getting another round to make sure you’re fully immunized… these people live in an alternate universe.
Don’t be the last one to figure out this scam.
Anyone still wants to get any more shots ? I surely wouldn’t want another round of them !
Onewhoquestions, I agree.
Dr. Hulda Clark said that every case of cancer she was familiar with was caused by a toxin and a parasite (bacteria,virus, fungal organisms included as parasites) Kind of sounds like the recipe for a proper vaccine.
Its good to be poisoned! We must poison you with Aluminum! Do you hear? Do you see? Healthy Food makes you Healthy. Poison makes you sick! Aluminum, Baby DNA, Formaldehyde, etc. Do you know the ingredients of vaccines?
If the infection stay at the injection sight you are very blessed if it go systemic . Than what ? Who pays for that clean up? And what happens if that bacteria can’t be cleaned up? I very tried of hearing that vaccines are safe when clearly there is always risk and side affects with all pharmaceuticals. Vaccines are not the exception period. And science is never settled it’s constantly evolving as we learn more. That’s why we must fight for freedom of choice over what goes into our bodies. As did our fathers did in WWII . This is not Germany this is the United States of America land of the free and we need to keep it that way!
Look at the vaccine media & excipients summary on the DCD website. The levels of aluminum have DOUBLED. Even my veterinarian has started testing dogs & cats hair for aluminum because of all the side effects from so many shots; especially tumors at the injection site.
Sorry that was CDC.gov.
Big pharma = witchcraft
Don’t be left out! Get yours today! HaHAHAHAHAHA! The good doctor should be the first one in line! They are, after all, completely safe. You could die if you don’t get one! You might kill your neighbor if you don’t get one! Do you want that kind of guilt on your hands? According to Darwin, all the people involved in this situation will eventually die out.
calling a spade a spade …. vaccination = infection by injection … but there is nothing to see here because vaccines are safe and everyone should have them … get your damn vaccines and never mind ANY of the facts that suggest otherwise
“Health department says Kentucky-based vaccine provider caused infections.” No, the provider didn’t cause infections … the VACCINES CAUSED INFECTIONS. period.
drink the kool-ade
All vaccines are contaminated from the start. If you want a hit you in the gut awakening on vaccines read: Fear Of The Invisible by Janine Roberts. Here are a couple of paragraphs from the chapter:
MMR Contaminated:
It was thus a shock to discover from this top-level scientific workshop that the viruses in our current vaccines are not in a sterile fluid as I had presumed, but in a soup of unknown bits and pieces, a veritable witches’ brew of DNA fragments, added chemicals, proteins and, even possibly prions and oncogenes, all of which would easily pass through the filters used to be injected into our children.
Our vaccines, I thus learnt, are not filtered clean but are suspensions from the manufacturers’ ‘incubation tanks’ in which the viruses are produced from ‘substrates’ of mashed bird embryo, minced monkey kidneys or cloned human cells. These suspensions are filtered before use but only to remove particles larger than viruses. The point of the vaccine is that it contains viruses, thus these must not be filtered out. This means there remains in the vaccine everything of the same size or smaller, including what the manufacturers call ‘degradation products’ – parts of decayed viruses or cells.
I also learnt that the only official checks made for contaminants in vaccines are for a few known pathogens, thus ignoring a vast host of unknown, unstudied, small particles and chemicals. These eminent doctors reported at these vaccine safety meetings that it is simply impossible to remove these from our common vaccines – and this would of course also apply to vaccines for pets, farm animals and birds.
I went to the published reports of the MMR manufacturers and found these confirmed what the scientists at this workshop had reported. A manufacturer stated in 2000 that it made the MMR vaccine with ‘harvested virus fluids.’ It stated frankly that their ‘Measles vaccine bulk is an unpurified product whose potency was measured through a biological assay for the active substance rather than through evaluation of integrity of physical form. Degradation products are neither identified nor quantified.’ In other words, it left the latter in the measles vaccine along with all contaminants that lay there quietly, or worked slowly. The pharmaceutical company admitted checking the measles vaccine only for obviously active contaminates. It did not measure how much the vaccine was polluted with genetic code fragments, other viruses, or with parts of bacterial, animal, bird or human cells.
Roberts went on a radio show and read how the MMR is manufactured. When the WHO got wind of it they created a new MMR report and made sure all pertinent websites got copies of it and they removed the old report. Fortunately Roberts had copied the genuine one and put it in her book.
I got non paralytic polio from too much vaccine when I was about to go in 2nd grade. I have R A now and I’ll bet vaccinations had everything to do with this.
we stopped any and all vaccines after my daughter’s booster tetanus. She ended up with a severely swollen leg, unable to move or walk for several days. My other child had a lesser reaction which the establishment would deem normal. Enough of that! Those who are in favor of these poisons can have all of ours, some one please make sure they get them!
What are we doing? The world has gone MAD! Did anyone happen to think as an after thought, “I wonder what is in the serum?” Did you read the insert? Did anyone read it? WOW! Have we been brainwashed to not question their “authority”? Don’t you dare question their authority. How long will this madness continue?
We had an outbreak of spinal meningitis in my company in the military. Yes! It was right after a barrage of medical “treatment” needles and airgun invasion to our bio systems!! In my world of physics and common sense. Puss, heavy metals and poisons chemicals are not conducive to health.
Of course you have to have a “Practitioners Degree” to conduct such insane “practices” Notice: I did not call them doctors. In the very early history of mankind, the Doctor was a teacher, not a drug pusher. In the early historical records this madness was referred to as pharmacia and sorcery. Early Greek.Yes! How long will this madness continue to plague our world?
“Once She Ran With Rivers Pure”. Please see: “Certain drugs, can cause meningitis.” https://www.cdc.gov/meningitis/index.html