
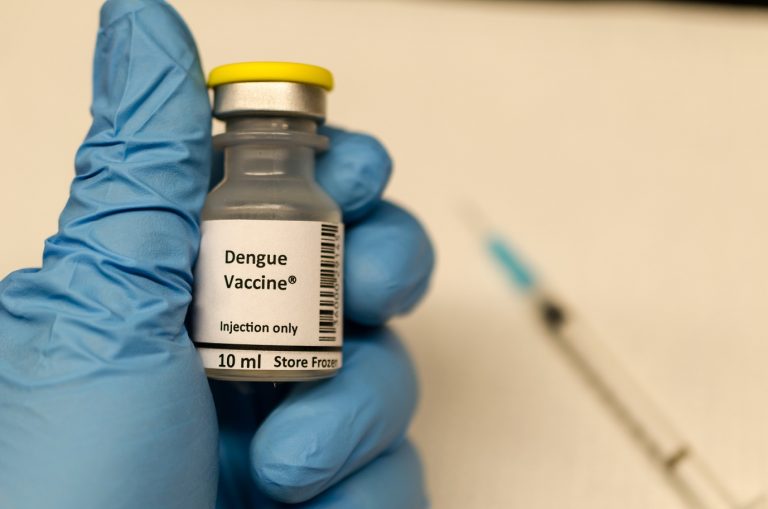
Sanofi Pasteur announced that its dengue vaccine, Dengvaxia, may lead to more severe cases of dengue fever in some people—a warning it relayed after the vaccine was already administered to more than 700,000 children in the Philippines. Those at risk, according to Sanofi, are people who have not been previously infected by dengue virus.
Their new analysis that evaluated long-term safety and efficacy of the vaccine revealed that “in the longer term, more cases of severe disease could occur following vaccination upon a subsequent dengue infection.”1
As a result, the Philippines has launched an investigation and convened an expert panel to figure out what to do next. Health Secretary Francisco Duque said in a news release, “The safety of the children vaccinated is paramount, and the Health Department will need to do surveillance of those given Dengvaxia with no prior infection. It’s really a big task.”2 Nearly 4 billion people across 128 countries are at risk of infection with dengue, a viral disease spread by mosquitoes.3
Symptoms of dengue fever range from mild (or none at all) to high fever along with headache, body pain, nausea, vomiting and pain behind the eyes, with most people recovering within a week.4 A severe form of the disease, known as dengue hemorrhagic fever or severe dengue, however, is life-threatening and can lead to difficulty breathing, circulatory system failure and death. It’s this severe form of dengue that Dengvaxia vaccine may increase risks for in people who have not previously been infected with dengue.
Sanofi Claimed Long-Term Safety Studies Were Completed, Now Recommends Against Vaccination in Some
In 2015, the Philippines’ Food and Drug Administration approved Dengvaxia, the first dengue vaccine in Asia, manufactured by Sanofi Pasteur. The vaccine was intended to prevent all four dengue types in individuals from 9 to 45 years of age living in endemic areas.5 At the time, Sanofi claimed studies had been conducted “affirming the vaccine’s consistent efficacy and longer-term safety profile in a study population 9 to16 years of age.”6
The new analysis, however, is based on up to six years of clinical data evaluating the long-term safety and efficacy of Dengvaxia in those who have been infected with dengue and those who have not. After the finding that people with no prior infection may be at increased risk of severe dengue if they receive Dengvaxia, Sanofi is proposing to update the vaccine label to request that health care professionals “assess the likelihood of prior dengue infection in an individual before vaccinating.”7
To date, none of the children who received Dengvaxia were tested for prior dengue infection. In fact, the current test available can only test for evidence of prior infection with all flaviviruses, including Zika, chikungunya and Japanese encephalitis, and would turn positive for exposure to any of them, including dengue.
As a result, health reform advocate and former past president of the Philippine College of Physicians, Dr. Anthony Leachon, told ABS-CBN News, “It means some of them will develop severe dengue, we don’t know who. All of them will have to live with this possibility for the rest of their lives.”8 Sanofi also proposed changing Dengvaxia’s label to state:
“Vaccination should only be recommended when the potential benefits outweigh the potential risks (in countries with high burden of dengue disease). For individuals who have not been previously infected by dengue virus, vaccination should not be recommended.”9
Philippine Government Purchased 3 Million Doses of Dengvaxia, Worth Nearly $70 Million
In April 2016, just four months after Dengvaxia’s approval, the Philippine government launched a school-based dengue vaccination program among fourth-graders in public schools in three regions hardest hit by dengue. Three million doses of the vaccine had been purchased by the government, worth nearly $70 million, and were intended to vaccinate 1 million schoolchildren (with three doses each). At the time, critics expressed concern about the vaccine program’s safety, since the vaccine was still being studied.
Some also questioned why the Philippine Department of Health (DOH) purchased so much of the vaccine in haste, since fewer than 730,000 students met the criteria for vaccination and, reportedly, only about 534,000 student’s parents gave informed consent for vaccination.10
The DOH-Dengue Vaccine Implementation Committee (DVIC) wrote in April 2017, “After allocating the vaccines … we have projected a large excess supply of Dengue vaccine.”11 While there was initially talk of expanding the vaccine program, Sanofi’s announcement likely will change that.
Speaking out on Facebook, former health undersecretary Susan Pineda Mercado described the dengue vaccine push as the “biggest government funded clinical-trial-masked-as-a-public-health-program scam of an experimental drug in the history of the DOH,” adding, “This was reckless and irresponsible from the start and the public was deceived into thinking this vaccine would protect children from dengue. The public health community has been outraged for over a year. Legal action is now necessary.”12
Meanwhile, prior to their latest announcement, Sanofi was working hard to make Dengvaxia a household word in the Philippines, even going so far as to air TV and radio advertisements for the vaccine. Unlike in the U.S., this type of direct-to-consumer advertisement of prescription drugs in the mass media is illegal in the Philippines. In December 2016, the Philippine FDA ordered Sanofi to stop the ads, while also asking TV and radio outlets not to air them.13
WHO Report Raised Red Flags About Dengvaxia in 2016
The World Health Organization (WHO) is among those in the midst of reviewing data on Dengvaxia following Sanofi’s announcement. However, the organization issued a report in 2016 that revealed “vaccination may be ineffective or may theoretically even increase the future risk of hospitalized or severe dengue illness in those who are seronegative at the time of first vaccination regardless of age.
If this is the case, even in high transmission settings there may be increased risk among seronegative persons despite a reduction in dengue illness at the population level.”14
Singapore’s Health Sciences Authority also identified risks of the vaccine upon its October 2016 approval, and reportedly was adding risk warnings to the drug’s label. In Brazil, where a public vaccination program with Dengvaxia was also implemented, health officials have changed recommendations to advise people who have never been infected with dengue to not receive the vaccine.
To date, three deaths have been reported among children who received Dengvaxia,15 but officials have so far said they were “coincidental” and not related to the vaccine.16
Findings Highlight the ‘Complex Nature of Dengue Infection’
In their statement, Sanofi said their new findings highlight the “complex nature of dengue infection,”17 which appears to be their way of trying to pass the buck for creating a vaccine that may lead to severe disease in some children. It’s also important to note that dengue is in no way unique in its complexity; other viral diseases exhibit similar complexity.
Yet, despite huge gaps in scientific knowledge about infectious microbes that cause disease, vaccine makers continue to create vaccines that attempt to outsmart the microbes, and typically fail, which often leads to severe consequences like with Dengvaxia.
It’s also not the first time that vaccination has worsened disease severity. For instance, in 2012 it was revealed that the 2008-09 flu vaccine was associated with increased risk for more serious H1N1 illness. Further, research published in the Journal of Virology also confirmed that the seasonal flu vaccine may actually weaken children’s immune systems and increase their chances of getting sick from influenza viruses not included in the vaccine.18
In addition, when blood samples from 27 healthy, unvaccinated children and 14 children who had received an annual flu shot were compared, researchers found that the unvaccinated children naturally built up more antibodies across a wider variety of influenza strains compared to the vaccinated group.
Another issue often disregarded in discussions of vaccine safety is the spread of vaccine-strain virus infections, including polio, along with widespread vaccine failures among vaccines for mumps, meningococcal disease and pertussis.
The conventional solution to failing vaccines is to add booster shots to the schedule, with unknown consequences for natural immunity and human health. It’s a complex situation indeed, which is why open conversations and unbiased research into vaccine safety and efficacy are so badly needed before a one-size-fits-all vaccine approach should be continually recommended.
Note: This article was reprinted with the author’s permission. It was excerpted and slightly modified from an article published on Dr. Mercola’s website at www.mercola.com.
References:1 Sanofi Pasteur November 29, 2017.
2 ABS CBN News November 30, 2017.
3 WHO, Dengue and severe dengue April 2017.
4 Mayo Clinic, Dengue Fever.
5 Sanofi Pasteur December 22, 2015.
6 Ibid.
7 See Footnote 1.
8 See Footnote 2.
9 See Footnote 1.
10 ABS CBN News June 21, 2017.
11 Ibid.
12 Rappler November 30, 2017.
13 Fox News January 3, 2017.
14 WHO Dengue Position Paper July 2016.
15 Reuters December 4, 2017.
16 See Footnote 10.
17 See Footnote 1.
18 Journal of Virology 2011 Nov;85(22):11995-2000.












2 Responses
So, less than 2 years of “after-market testing” revealed the dangers of this vaccine that was not detected in 6 years of pre-market clinical testing. Hmmm……. sloppy work.
There may also be a correlation with arthritis, just like the parvo vaccine. My pet had lameness after a vaccine. The doctor then wanted $900.00 to figureout why with new labs and Xrays. They make a lot of money and hurt out pets unnecessarily. Read about Banfield’s computer system and how they dumb down the doctor. The pet vaccines are not adjusted for weight either.