FDA Revokes EUA Status for COVID-19 Shots

The U.S. Department of Health and Human Services (DHHS) has revoked emergency use authorization (EUA) for the COVID-19 shots. The federal government had relied on the EUA status to justify release of the COVID-19 biologicals in December 2020 for public distribution without full licensure by the FDA.1 On Aug. 27, 2025, DHHS Secretary Robert F. […]
Expired COVID-19 Shots Repurposed for Biosensor Research

During the COVID-19 pandemic, pharmaceutical companies manufactured mRNA COVID-19 shots in massive quantities. While many of the shots were distributed and administered, a large number went unused, eventually expiring and being discarded. To avoid this type wastage in the future, researchers at the Center for Physical Sciences and Technology (FTMC) in Lithuania are proposing a […]
Government Agencies Ordered to Erase All Federal Employee COVID Vaccination Records

U.S. government agencies have been ordered to erase any records of employees’ COVID-19 vaccination status, noncompliance with pandemic-era mandates, or vaccine exemption requests, according to an Aug. 8, 2025 memo from U.S. Office of Personnel Management (OPM) Director Scott Kupor. Issued in response to recent litigation, the directive comes four years after the previous administration […]
Thimerosal to Be Banned from Use in All Vaccines in U.S.
In 1999, the U.S Public Health Service, along with the American Academy of Pediatrics (AAP) and vaccine manufacturers, jointly agreed that any potential risk from thimerosal, a mercury-based preservative found in some inactivated vaccines, warranted its removal as soon as possible. Over the next few years, most routine pediatric vaccines in the U.S. became thimerosal-free […]
Dental Floss Tested as a New Route for Vaccine Administration
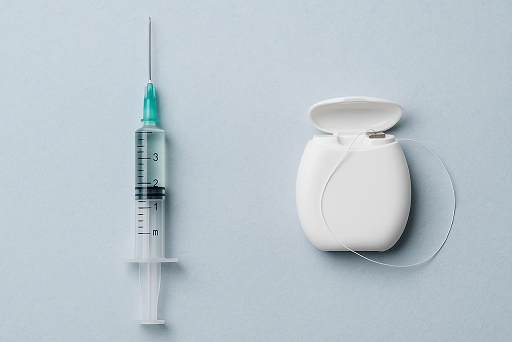
In a 2025 study published in Nature Biomedical Engineering, researchers reported that applying floss laced with vaccine components such as proteins and inactive viruses along the gum lines of mice triggered an immune response.1 The goal is to use dental floss to vaccinate large numbers of people during a pandemic. The researchers found that “Floss-based […]
Pre-filled Syringe Version of GSK’s Shingles Vaccine Approved by FDA
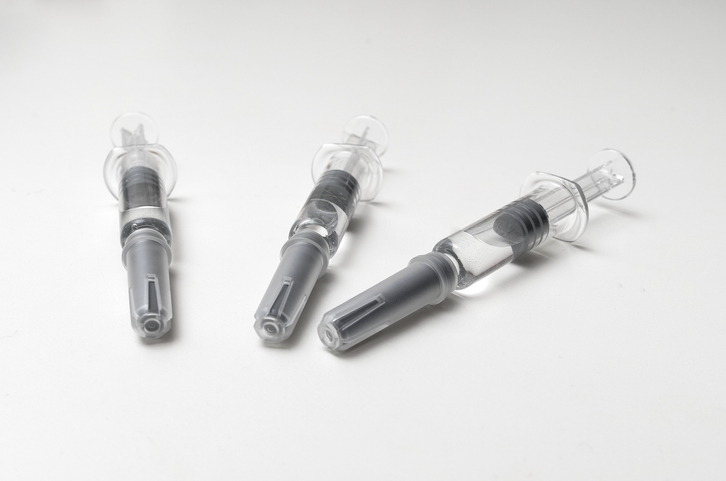
On July 17, 2025, officials at the U.S. Food and Drug Administration (FDA) granted a license to GSK plc (formerly GlaxoSmithKline) to distribute a pre-filled syringe version of Shingrix (Zoster Vaccine Recombinant Adjuvanted) vaccine to control outbreaks of shingles (herpes zoster) in adults. The pre-filled syringe version of Shingrix was approved for use by adults […]
