Merck and Pfizer Seek Approval for COVID-19 Antiviral Drugs

Merck and Pfizer have submitted Emergency Use Authorization (EUA) applications to the U.S. Food and Drug Administration (FDA) to authorize distribution of what would be the first antiviral drug specifically designed to treat COVID-19 disease.1 Merck/Ridgeback Biotherapeutics are seeking EUA approval for Molnupiravir (MK-4482/EIDD-2801), an investigational oral antiviral drug originally developed to treat influenza.2 Pfizer […]
FDA Asks Court to Delay Full Release of Documents on Pfizer COVID Biologic for 55 Years

In response to a Freedom of Information Act (FOIA) lawsuit filed on Sept. 16, 2021 against the U.S. Food and Drug Administration (FDA) by a medical transparency group of more than 30 physicians, professors and scientists from various universities in the United States, the FDA has asked a federal judge to delay the full release […]
FDA, CDC Green Light Pfizer’s Experimental COVID Biologic for Children 5 to 11 Years Old
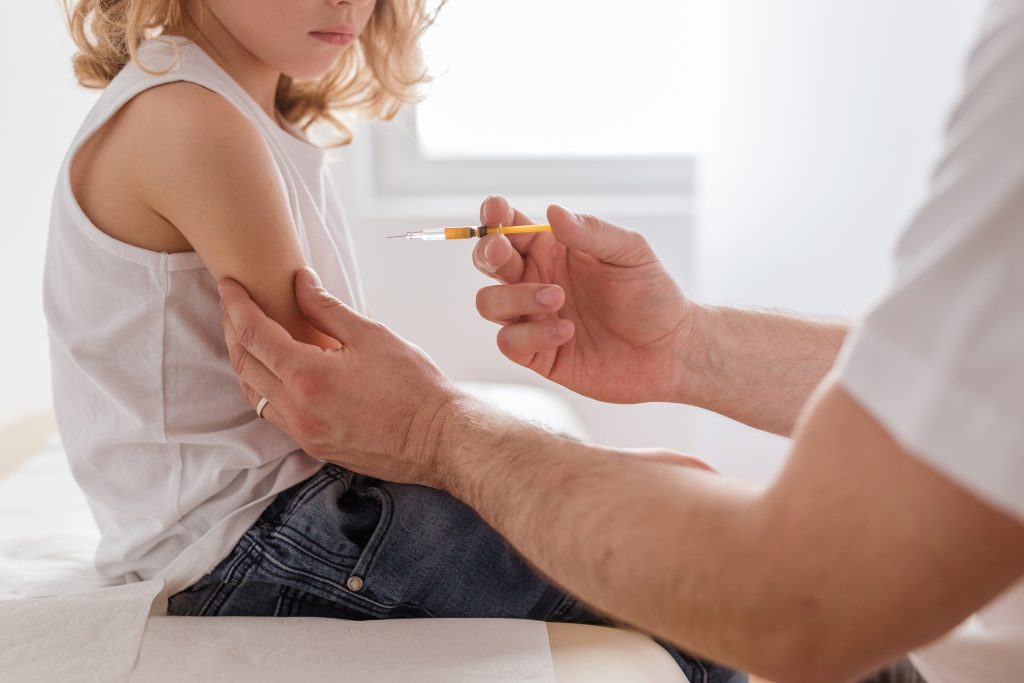
One Oct 29, 2021, the U.S. Food and Drug Administration (FDA) granted Emergency Use Authorization (EUA) for the experimental messenger RNA (mRNA) Pfizer/BioNTech COVID-19 biologic BNT162b2 for children five to 11 years old. The FDA’s decision, which is based on a clinical trial earlier this year involving 2,268 kindergartners and elementary school-aged kids and an […]
Senators, Doctors Urge CDC to Recognize Natural Immunity

As millions of Americans have experienced COVID-19 disease and the COVID vaccine mandate issued by the government continues to impact the lives of the majority of working Americans, Senator Roger Marshall, MD (Kansas), along with several other congressional Doctors Caucus members, are urging Center for Disease Control and Prevention (CDC) director Rochelle Walensky, MD to […]
White House Reveals Plans to Give COVID Shots to All 5 to 11 Year Old Children

On Oct. 20, 2021, the Biden administration outlined plans to begin giving COVID-19 vaccinations to children between five and 11 years old. Pending the results of reviews of the plan by the U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) over the […]
Nordic Nations Suspend Use of Moderna COVID Shot Over Heart Inflammation Concerns

Following reports of an increased risk of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the lining of the heart) in young adults after receiving Moderna/NIAID’s experimental messenger RNA (mRNA) COVID-19 biologic known as “Spikevax,” the governments of Denmark, Norway and Sweden have suspended the shot or will not make it available for […]
