Idaho Health Department Bans Giving COVID-19 Shots
In Idaho, the Southwest District’s Board of Health has voted to prohibit COVID-19 shots from being administered in its medical facilities, a regional public health department in Idaho, has been prohibited from administering COVID shots to residents in six counties along the Idaho-Oregon border.1 The regional public health department serves Adams, Canyon, Gem, Owyhee, Payette […]
Canadian Man Euthanized After Diagnosis of ‘Post COVID-19 Vaccination Syndrome’
A man in his late 40s from Ontario, Canada, who had a history of mental illness, underwent euthanasia under the Medical Assistance in Dying (MAID) program, after program assessors concluded that his physical decline was most reasonably attributed to post COVID-19 “vaccination syndrome” that developed after he received three COVID shots. The term remains controversial, […]
Man Suffers Painful Facial Nerve Damage After Pfizer’s mRNA COVID-19 Booster Shot

A 54-year-old man from the Omagh, Northern Ireland suffered a severe adverse reaction just days after receiving the Pfizer/BioNTech’s Comirnaty mRNA (messenger ribonucleic acid) COVID-19 booster shot in December 2021. Larry Lowe, a former college lecturer, musician and runner, said he developed numbness on the right side of his face and began to experience severe […]
Influenza-COVID Combo Vaccine Trial Halted Over Safety Concerns

The U.S. Food and Drug Administration (FDA) has put a hold on Novavax’s COVID-influenza combination vaccine and standalone influenza vaccines being tested in clinical trials after a participant reported nerve damage following receipt of the experimental vaccine in January 2024.1 Reported symptoms were diagnosed as motor neuropathy, a disease process that can be acquired, hereditary […]
WHO Endorses a New Single-Dose HPV Vaccine
The World Health Organization (WHO) announced on Oct. 4, 2024 that it has prequalified its fifth human papillomavirus (HPV) vaccine, Cecolin. Cecolin is the only vaccine approved for a single-dose regimen, setting it apart from multi-dose HPV vaccine options like Merck’s Gardasil vaccine, which typically requires three doses, and Walrinvax vaccine, which is manufactured by […]
Man’s Face Paralyzed After Receiving Pfizer’s COVID Shot
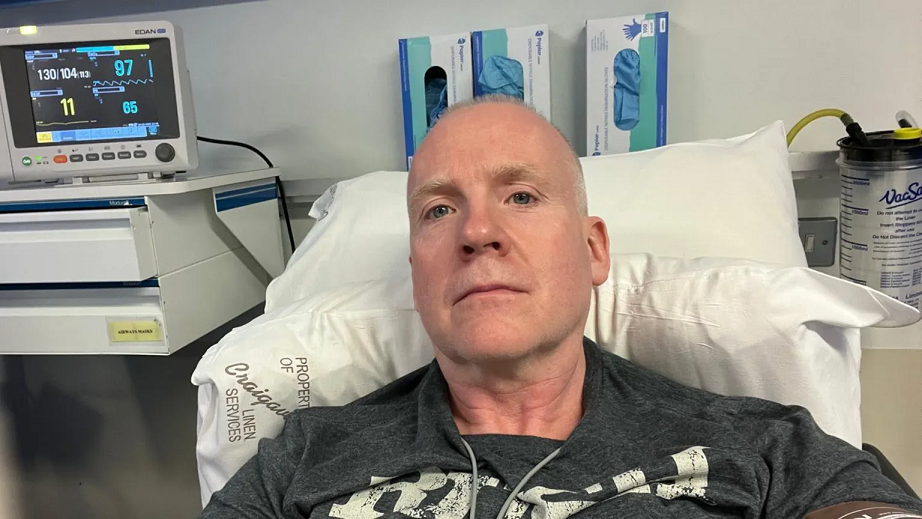
Larry Lowe, a 54-year-old resident of Omagh, Northern Ireland, experienced facial paralysis after receiving the Pfizer/BioNTech’s Comirnaty mRNA COVID-19 shot on Dec. 15, 2021. Prior to getting vaccinated, Lowe was healthy and physically active. Shortly after he receiving the shot, he developed numbness on the right side of his face and persistent pain. “I had […]
